Trends and disparities in heart failure mortality with and without chronic kidney disease in a nationwide retrospective analysis
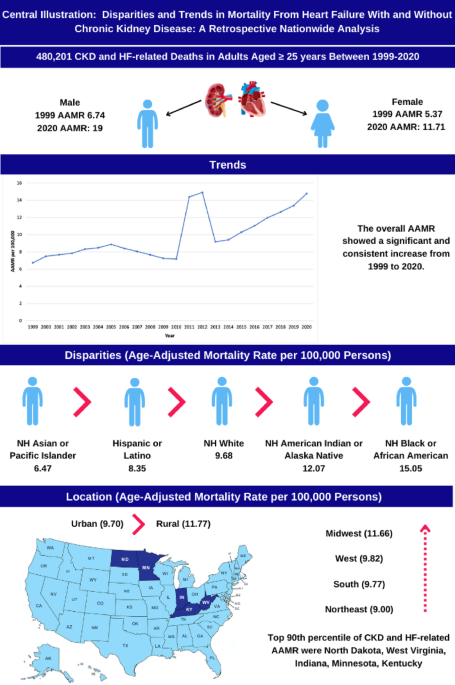
Our analysis of mortality data from the CDC WONDER database, spanning two decades, reveals several key findings. First, we identified a steady and significant increase in mortality related to concurrent HF and CKD in the U.S. from 1999 to 2020. This upward trend is evident in both men and women, with men consistently showing higher rates compared to women. Second, clear ethnoracial disparities were noted, with NH Black individuals experiencing the highest concurrent HF and CKD-related mortality rates compared to other ethnoracial groups. Third, we found significant regional disparities in HF and CKD-related mortality, with the Midwest showing the highest rates relative to other regions. Rural areas also exhibited higher mortality rates than urban areas. Lastly, the rate of mortality for concurrent HF and CKD was higher than HF or CKD alone.
CKD is linked to a significantly increased risk of CVD and CV-related mortality. Studies involving CKD patients have shown a higher relative risk of coronary heart disease, HF, and stroke compared to individuals without CKD10. CV mortality among CKD patients is considerably higher than in the general population, with left ventricular hypertrophy and HF contributing to the poor prognosis25. Both conditions show rising incidence and prevalence in older age groups, as well as among individuals with hypertension, diabetes mellitus, or other risk factors for cardiovascular and kidney diseases26. The presence of one condition often accelerates the onset and progression of the other, and having both significantly increases the risk of hospitalization, rehospitalization, need for intensive care or kidney replacement therapy, and mortality27,28,29. A study from the Acute Decompensated Heart Failure National Registry (ADHERE) involving 105,388 HF patients found that 57% had coronary artery disease, 73% had hypertension, 44% had diabetes, and over 60% had kidney disease30. These patients had a mean age of 72 years, with 20% being Black individuals and 52% women30. Age is a major risk factor for HF and its associated conditions, including CKD. Consequently, the prevalence of CKD is expected to be higher in HF patients than in the general population31. Furthermore, Dhingra and colleagues demonstrated that individuals with an estimated glomerular filtration rate (eGFR) of < 60 mL/min per 1.73 m² had a twofold higher risk of developing HF compared to those with an eGFR ≥ 60, based on a cohort of 10,181 participants followed over a 10-year period32. This highlights the urgent need to raise awareness about the high prevalence of HF in CKD, particularly in advanced stages (stages 4–5 and those requiring kidney replacement therapy), and to implement effective treatment strategies33.
Our analysis shows a significant increase in mortality for both sexes over the study period, with males exhibiting substantially higher mortality rates compared to females. This elevated mortality in men is attributed to higher rates of CV risk factors such as hypertension and metabolic syndrome, which are exacerbated in CKD34. Men often present with more advanced CKD and start treatment later, leading to poorer outcomes34,35. Biological differences, including sex hormones and body composition, also contribute, with men showing higher rates of atherosclerosis and CV events36,37.
Observational studies suggest that the decline in GFR occurs more rapidly in men than in women, particularly in unadjusted analyses of middle-aged and elderly individuals38,39,40. A cohort study by Ricardo et al. found that women had a significantly lower risk of ESRD, a 50% decline in eGFR, progression to CKD stage 5, and death38. Additionally, Melsom et al. demonstrated that among middle-aged and elderly individuals in the general population, the mean GFR decline was slower in women than in men, independent of overall health status39. Further, men appear to be more susceptible to hypertension-related GFR decline, as observed in a Chinese study, and African-American men with early CKD exhibit poorer hypertension control compared to women41,42. Ambulatory blood pressure (BP) monitoring has also shown that men with CKD (stages 2–5) have higher daytime and nighttime systolic BP than women, which may be a key contributor to their increased risk of adverse outcomes43. These observations may be partially explained by the role of sex hormones. The lower prevalence and slower progression of CKD in females may be attributed to estrogen’s protective effects44. Recent studies indicate that estrogen receptors are present not only in reproductive organs but also in solid organs such as the brain, liver, and kidneys, suggesting a direct physiological role44. Activation of estrogen receptor α in the renal vasculature has been shown to stimulate nitric oxide synthase via a phosphoinositide 3 kinase–Akt–dependent pathway, promoting vasodilation45. Additionally, activation of estrogen receptor β attenuates inflammatory responses induced by tumor necrosis factor α in vascular smooth muscle cells, further contributing to renal protection in females46.Conversely, androgens may have detrimental effects on kidney function. Animal studies have demonstrated that age-dependent glomerular damage is more pronounced in male rodents, while castrated males are protected from such injury, suggesting a harmful role of androgens47. Potential mechanisms include genetically determined sex-based differences in renal structure and function, receptor-mediated effects of sex hormones on glomerular architecture, and influences on the synthesis and release of cytokines, vasoactive agents, and growth factors, all of which may contribute to increased CKD progression and mortality in men48. Paradoxically, despite the slower progression of CKD in women, men constitute the majority of patients initiating renal replacement therapy (RRT). This discrepancy may be attributed to the protective effects of estrogen in women and/or the detrimental effects of testosterone, coupled with unhealthy lifestyles in men, which accelerate kidney function decline. Additionally, elderly women are more likely to opt for conservative management rather than RRT, further influencing these observed differences49.
Conversely, literature suggests that women with CKD may experience more severe cardiovascular outcomes in certain contexts, with reduced eGFR and albuminuria having a pronounced impact34. In the general population, female life expectancy is longer than male, and the risk of CV events is higher in men than women, possibly due to sex hormones50. In CKD, altered sex hormone production may reduce the protective effect of female sex on CV events. Observational studies in CKD cohorts often show higher CV morbidity and mortality in men compared to women. However, when comparing mortality rates, women with CKD have a higher CV burden than age-matched women in the general population, and men with CKD have a higher CV burden than men in the general population34. This necessitates sex-specific approaches in concurrent HF and CKD-management to address differing risks and outcomes.
Our findings also show notable ethnoracial disparities, with NH-Black populations having the highest concurrent HF and CKD-related mortality rates and young NH Asian or Pacific Islander populations having the lowest. Black individuals have a higher prevalence of CKD and CV events, partly due to genetic variants (e.g., APOL1 risk alleles) and higher rates of hypertension and obesity35,51,52,53,54,55. Gutiérrez et al. reported that around 13% of Black individuals in the U.S. possess a high-risk APOL1 genotype, which is linked to a higher prevalence and faster progression of CKD compared to other racial groups, contributing to a disproportionate CKD burden56.
Multiple case-control studies have established a strong link between the G1 and G2 APOL1 risk alleles and various kidney diseases in African Americans. Genovese et al. found that individuals carrying two APOL1 risk alleles had over 10-fold increased odds of developing focal segmental glomerulosclerosis and 7-fold increased odds of hypertension-related ESRD57. Furthermore, among 1,776 African Americans in the Dallas Heart Study, those with two APOL1 risk alleles had a three- to fourfold increased odds of prevalent microalbuminuria and reduced estimated GFR (eGFR < 60 ml/min per 1.73 m²) compared to those with zero or one risk allele. These associations varied based on diabetes status, with no significant correlations observed in non-diabetic participants58. The APOL1 gene encodes apolipoprotein-1, a trypanosome lytic factor found in human serum and a component of HDL particles59. A recent biopsy study identified apolipoprotein-1 within the smooth muscle cells of medium-sized arteries and arterioles in patients with focal segmental glomerulosclerosis or HIV-associated nephropathy but not in normal kidney samples60. This suggests that APOL1 variants may contribute to kidney disease progression through arterial wall damage and vascular dysfunction. Additional potential mechanisms include APOL1 variant effects on lipid metabolism or autophagy61,62. Additionally, Black and Hispanic populations have higher rates of diabetes, hypertension, and obesity, major factors driving their elevated CKD burden relative to other racial groups63. A multiethnic study by Bansal et al. identified that CKD was associated with an increased risk of HF, similar in magnitude to the risk of coronary heart disease and greater than that of stroke10. This elevated risk was particularly pronounced in Black and Hispanic individuals. Additionally, a related study by Domingo et al. found that the link between kidney dysfunction and HF was stronger in Black individuals compared to White individuals64. The impact of APOL1 gene variants on kidney disease is significantly influenced by social determinants of health (SDOH), exacerbating disparities in CKD progression among African Americans. Limited access to healthcare, poor hypertension control, and racial biases in treatment contribute to worse outcomes for APOL1 high-risk individuals.
Furthermore, Black and Hispanic populations have higher rates of diabetes, hypertension, and obesity, major factors driving their elevated CKD burden relative to other racial groups63. A multiethnic study by Bansal et al. identified that CKD was associated with an increased risk of HF, similar in magnitude to the risk of coronary heart disease and greater than that of stroke10. This elevated risk was particularly pronounced in Black and Hispanic individuals. Additionally, a related study by Domingo et al. found that the link between kidney dysfunction and HF was stronger in Black individuals compared to White individuals64. Black patients have the highest risk of HF-related death65. Further, the rate of HF hospitalizations for Black men and women is nearly two-and-half fold higher than the rate of HF hospitalizations for Whites, with costs that are significantly higher in the first year after HF hospitalizations66,67. While the relative rate of HF hospitalizations has improved for other race/ethnic minorities, the disparity in HF hospitalizations between Black and White patients has not decreased during the last decade66.
Despite the increased prevalence of advanced CKD among Black individuals, they are less likely than white individuals to receive nephrology care prior to initiating dialysis68. Black individuals are also more likely than white individuals to start dialysis with a catheter, rather than having an arteriovenous fistula surgically placed for optimal vascular access and are less likely to receive treatments such as home haemodialysis or peritoneal dialysis, both of which are associated with improved physical function compared with in-centre haemodialysis69,70. Black individuals are also significantly less likely than white individuals to undergo kidney transplantation; numerous barriers contribute to this disparity, including clinician bias and the use of complicated clinical transplantation evaluation processes71,72. Additionally, a 2021 study demonstrated that the use of the race coefficient in eGFR equations can contribute to delays in obtaining transplant waitlist priority among Black individuals73.
Moreover, psychosocial factors such as discrimination and stress further exacerbate health issues by promoting neuro-hormonal activation and inflammation34. Structural racism and care disparities profoundly impact Black patients with HF, who are less likely to receive timely ambulatory care, specialized cardiology services, or intensive care unit admissions74,75. NH Black individuals also face significant barriers to accessing outpatient clinics, initiating guideline-concordant therapy, and often depend more heavily on emergency departments compared to Hispanic and White adults75. Racial disparities in kidney disease progression and HF care are heavily influenced by social determinants like neighborhood segregation, food insecurity, and limited access to healthcare services76,77. Higher rates of uninsurance and inadequate management of disease risk factors disproportionately impact NH Black, Hispanic, and Latino populations, leading to delayed treatment and exacerbating inequities76,77,78.
With regards to age groups, the highest mortality was reported among individuals aged 65 and older, consistent with previous literature51,79,80,81,82. Aging naturally leads to a decline in kidney function, with a reduction in glomerular filtration rate (GFR) starting after age 40, accelerating after 6579,80,82. The prevalence of CKD is notably higher in this age group, with about 39.4% affected51. Age-related kidney changes, such as glomerulosclerosis and interstitial fibrosis, significantly impact CKD progression. Older adults face elevated risks of HF due to age-related factors like systolic dysfunction, diabetes, and ischemic heart disease51,79. Additionally, frailty in the elderly exacerbates CKD outcomes, leading to increased vulnerability to complications and reduced survival rates79,80. The CKD-REIN study underscores that older CKD patients face higher CVD rates, including both atheromatous and non-atheromatous conditions83. Furthermore, the increasing prevalence of diabetes, obesity, and hypertension among younger adults, combined with potentially inadequate management and delayed diagnoses, has also intensified concurrent HF and CKD-related mortality rates in this stratum51.
Our findings indicate that rural areas have a higher burden of concurrent HF and CKD-related mortality compared to urban areas. Particularly from 2010 onward, the mortality gap between rural and urban areas has widened84. Rural Americans often face lower socioeconomic status, an older population, and reduced healthcare access53. Higher rates of risk factors such as smoking, obesity, and sedentary lifestyles are prevalent in rural settings. Workforce shortages and transportation barriers such as longer travel distances further complicate concurrent HF and CKD management, leading to poorer outcomes53. From 2002 to 2015, the decline in primary care physicians in rural regions occurred at twice the rate seen in urban areas. The widespread closure of hospitals over the last decade, especially during the COVID-19 era has further exacerbated these issues85. Furthermore, a study conducted by Crouch et al. revealed that rural counties with high rates of CKD, diabetes, and blood pressure, deemed high need, were less likely to have a highly rated dialysis facility, thereby potentially contributing to a higher mortality rate86.
Among census regions, the Midwestern region exhibited the highest mortality burden as compared to other regions. This may be due to inconsistencies in outpatient cardiology practices, such as the implementation of guideline-directed therapy, the influence of state regulations on Medicaid, limited access to quality healthcare, an increased burden of comorbidities, and a poor sedentary lifestyle87. Regional differences in mortality rates for HF hospitalizations reveal that the Midwest has the highest in-hospital mortality rates, while the Northeast has the lowest. These variations may reflect factors beyond hospital care quality, including disease severity, healthcare access, and regional patient or hospital characteristics88. The quality of in-hospital care is similar across regions, suggesting that regional disparities are influenced more by patient demographics, disease severity, and access to care than by treatment quality88. Furthermore, our analysis demonstrated higher mortality rates due to concurrent HF and CKD in states like North Dakota, West Virginia, Indiana, Minnesota, and Kentucky due to a combination of factors. These states have a high prevalence of risk factors such as hypertension, diabetes, obesity, and smoking89. Rural states like North Dakota and Kentucky face healthcare access barriers, including fewer facilities and specialists, leading to delayed diagnoses and inadequate management of concurrent HF and CKD, further exacerbated by limited access to nephrologists and cardiologists90,91. Additionally, states like Minnesota and Indiana have significant aging populations, who are more vulnerable to concurrent HF and CKD due to age-related organ decline and the cumulative burden of chronic diseases92,93. Socioeconomic disparities in these regions also contribute to poor disease management and outcomes, compounding the mortality rates.
With regards to specific policy recommendations, in order to enhance healthcare delivery for patients with concurrent HF and CKD, it is imperative to implement concrete policy interventions that address both systemic access issues and care integration strategies. Expanding access to nephrology and cardiology services in rural areas is crucial, as these regions often face shortages of specialized care, leading to delayed diagnoses and suboptimal management. Policy measures could include incentivizing specialists to practice in underserved areas, utilizing telemedicine to bridge geographical gaps, and establishing mobile clinics to provide on-site care94,95. Additionally, implementing integrated, person-centered care models has shown substantial benefits for patients with complex comorbidities. Regular monitoring and proactive management of modifiable cardiac risk factors, such as diabetes and hypertension, can help prevent the onset and slow the progression of CKD. This can be achieved through key best practices, including effective communication among healthcare providers, patient education and empowerment, and the early identification and management of CKD and HF symptoms13. A recent randomized trial highlighted that such integrated care approaches not only improved health-related quality of life but also showed a trend towards reduced HF hospitalizations, highlighting the potential of these models to enhance patient outcomes96.Moreover, implementing multidisciplinary teams comprising cardiologists, nephrologists, endocrinologists, and specialized nurses can facilitate comprehensive management of HF and CKD, ensuring coordinated care that addresses the multifaceted needs of these patients97. Furthermore, fostering collaborative care models that promote seamless communication among healthcare providers can mitigate the risks associated with fragmented care, ultimately leading to improved clinical outcomes and patient satisfaction98.
Managing patients with concurrent HF and CKD is particularly challenging, as they are more susceptible to the renal and metabolic adverse effects of HF therapies99,100. While β-blockers, renin-angiotensin-aldosterone system inhibitors, mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 inhibitors are well-established in HF with reduced ejection fraction, their safety and efficacy in advanced CKD remain less defined101,102,103.
Clinical trials such as Empagliflozin Outcome Trial in Patients with Chronic Heart Failure with Preserved Ejection Fraction (EMPEROR-Preserved) and Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure (DAPA-HF) have demonstrated that sodium-glucose cotransporter-2 inhibitors reduce cardiovascular mortality and HF-related hospitalizations, even in patients with moderate CKD103,104. However, concerns about hyperkalemia, worsening renal function, and hypotension frequently limit the use of RAAS inhibitors and MRAs in this population103,104. Given these complexities, a multidisciplinary approach involving both cardiology and nephrology is essential to optimize treatment, balancing the benefits of guideline-directed medical therapy with the risk of kidney function deterioration97. Emerging therapies, such as non-steroidal MRAs and novel vasodilatory agents, may provide additional options for patients with coexisting HF and CKD, underscoring the need for further research to refine treatment strategies99,105. Furthermore, a recent meta-analysis by Ahmed et al. demonstrated that Finerenone significantly reduced all-cause mortality, major adverse cardiovascular events, and heart failure-related hospitalizations106. While it showed a trend toward lowering cardiovascular death risk, it did not reach statistical significance106.
link