Regulatory considerations for successful implementation of digital endpoints in clinical trials for drug development
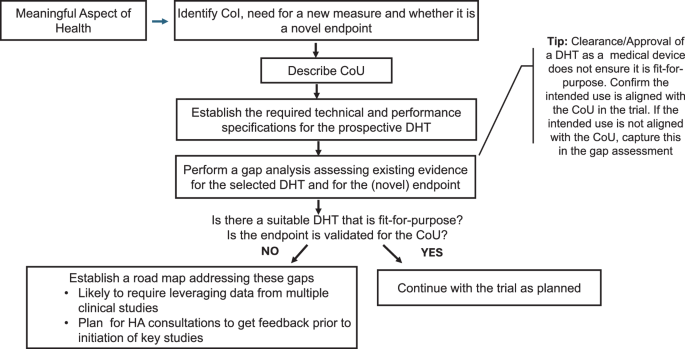
Advances in Digital Health Technologies (DHTs) have opened new opportunities in clinical trials by providing innovative methods to collect information from participants1. DHTs consist of hardware and/or software and can be used on various computing platforms, such as mobile phones, smartwatches, etc.2,3. More advanced DHTs also leverage artificial intelligence and machine learning for data processing and analysis4. The use of DHTs in clinical trials offers several potential advantages by providing richer data sets (e.g., continuous data collection in participant’s home environment vs. snapshots/episodic recordings during clinical visits) or performing objective measurements without recall bias that flaws Patient Reported Outcomes, leading to a better understanding of the efficacy and safety of the interventions studied1,3,5,6. Via remote data acquisition, DHTs can also decrease the number of clinical trial sites and follow-up visits and potentially increase diversity and inclusivity in clinical trials7. For these and other advantageous reasons, we have seen the use of DHTs in clinical studies increase significantly over the years1,8,9,10. Recently, the first example of digital outcome assessment used in a pivotal study was completed, the Phase III REBUILD clinical trial of INOpulse for the treatment of fibrotic interstitial lung disease, the primary endpoint was the change in moderate to vigorous physical activity, as measured by actigraphy (NCT03267108). Unfortunately, the study failed to meet its primary endpoint.
Regulators and legislatures have recognized the potential of DHTs in clinical trials and provided guidance and documents to help support the development of these new tools3,5,6,11,12. As part of the US Food and Drug Administration’s (FDA) work to fulfill the Prescription Drug User Fee Act (PDUFA) VII commitments, FDA has recently published the Framework for the Use of DHTs in Drug and Biological Product Development and established the DHT Steering Committee which consists of senior staff from the Center for Drug Evaluation and Research (CDER), the Center for Biologics Evaluation and Research (CBER), and the Center for Devices and Radiological Health (CDRH) to support the implementation of the framework for the use of DHTs in drug and biological product development12,13. Additionally, the US FDA has created the Digital Health Center of Excellence, based in CDRH, which is intended to provide scientific expertise on DHTs for all of FDA and connect internal and external stakeholders to FDA in the DHT space. To further this mission, the US FDA created the Digital Health Advisory Committee in October 2023 to advise the Commissioner of Food and Drugs on scientific and technical issues related to DHTs. As part of the PDUFA VII commitments FDA has also agreed to hold a series of five public meetings to discuss the use of DHTs in regulatory decision making and identify three demonstration projects to further progress the evaluation of DHTs.
The recent landmark qualification of the stride velocity 95th centile as a primary endpoint for ambulatory Duchenne Muscular Dystrophy studies by the European Medicines Agency (EMA) demonstrates the regulatory utility of DHT-derived endpoints in drug development and approval processes14. This endpoint is also currently under review by the US FDA as part of the Drug Development Tools, Clinical Outcome Assessment (COA) qualification pathway15,16. In addition, US FDA qualified the first DHT under the Medical Device Development Tools program, as a biomarker to help evaluate estimates of atrial fibrillation burden as a secondary effectiveness endpoint within clinical studies intended to evaluate the safety and effectiveness of cardiac ablation devices17. This qualification is for a specific context of use (CoU) in medical device development and is not directly applicable to clinical studies for drug development in atrial fibrillation.
Regulatory acceptance of DHTs can be a rigorous and lengthy process. It necessitates results from multiple prospective studies to demonstrate the validity and reliability of DHT, and clinical relevance of the DHT-derived endpoint1,3,6,18,19.
While this manuscript focuses on the regulatory considerations of using DHTs for capturing COAs and biomarkers in clinical trials, it is crucial to consider other important aspects such as technical, operational, and ethical considerations. These topics are beyond the scope of this manuscript, but readers are referred to previous publications9,20,21.
link