Implementation of Metformin Therapy to Ease Decline of Kidney Function in Polycystic Kidney Disease (IMPEDE-PKD): study protocol for a phase III, multi-centre, randomized, placebo-controlled trial evaluating the long-term efficacy of metformin in slowing the rate of kidney function decline in patients with autosomal dominant polycystic kidney disease | Trials

Explanation for the choice of comparators {6b}
Participants in the control group will receive matched identical placebo. A placebo-controlled trial was selected to rigorously evaluate the efficacy, safety and tolerability of metformin. The use of placebo as a comparator will increase the likelihood of detecting a treatment effect with a smaller sample size, thereby increasing research efficiency. The current justification for the use of a placebo comparator fulfils the criteria set out by the Declaration of Helsinki.
Intervention description {11a}
Because of the relatively high rate of gastrointestinal intolerance to immediate release metformin reported in pilot trials [17, 18] we chose treatment with a better tolerated extended release (XR) formulation of metformin [20].
At week 0 of the 12-week active run-in, all participants will start at the lowest dosage of metformin XR 500 mg tablets daily (taken with evening meal). This will be up-titrated every 4 weeks according to tolerability and kidney function. For participants with eGFR ≥ 45 mL/min/1.73 m2, the initial dose will be 2 × 500 mg up to a maximum of 2000 mg a day. Participants with eGFR values between38 and 44 mL/min/1.73 m2 will start at 500 mg daily up to a maximum of 1000 mg per day. The metformin XR dose deemed to be tolerated by a participant during the week 10 phone visit will be the participant’s starting dosage for the trial phase. An overview of dose titration during the run-in phase is provided in Fig. 2.
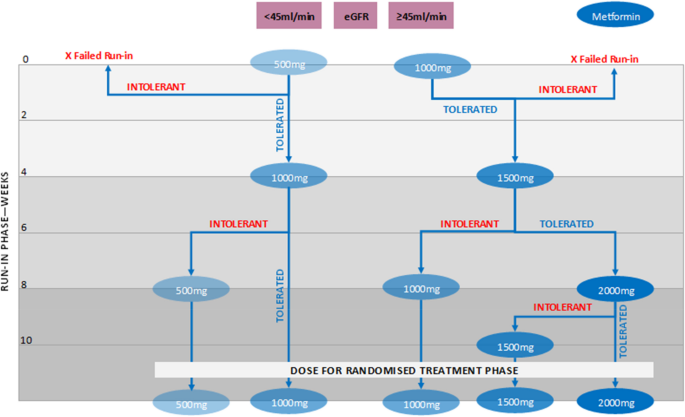
Study medication active run-in titration overview
The metformin prescribed during the run-in phase will be stopped by all participants at week 10 followed by a 2-week wash-out period. At the conclusion of the run-in phase (during the week 12 visit), participants will be randomly allocated to receive either metformin or matched placebo. Both those randomized to metformin and placebo will receive new tablets. Metformin and placebo will be provided in the same size 500 mg tablets; thus, participants will be taking between 1 and 4 tablets daily based on the dose tolerated and eGFR.
Baseline assessments for the trial phase will be conducted at the week 12 visit, which will also serve as month 0 of the trial phase.
Criteria for discontinuing or modifying allocated interventions {11b}
Medication tolerability will be adjudicated at each study visit by site investigators asking participants how they are tolerating the current dose and if they are experiencing any gastrointestinal side effects. If the dose is not tolerated, adjustments will occur in decrements of 500 mg. If a participant’s eGFR falls within the range of 30–44 mL/min/1.73 m2, the maximal dose will be reduced to 1000 mg/day.
The study medication may be temporarily or permanently discontinued if any of the following are present:
-
• A serious adverse reaction thought likely due to the study medication
-
•Requirement to prescribe study-prohibited medication or medication with known interactions with metformin (for example, cimetidine)
-
• Clinical indication for metformin (for example, a new diagnosis of T2DM)
-
• Inability to tolerate minimum dosage of study medication
-
• eGFR < 30 mL/min/1.73 m2 (as per FDA and TGA guidelines) for a minimum of two measurements in the same week
-
• Pregnancy or condition (as per treating clinician’s discretion) where continuation of the study medication is not considered in the participant’s best interests
-
• Uncontrolled hypertension as defined by systolic blood pressure > 160 mmHg or diastolic blood pressure > 100 mmHg after a period of rest, or
-
• Requested by the participant or their treating physician.
If participants are unable to maintain adequate fluid intake (e.g. due to gastrointestinal upset or dehydration), they will be advised to withhold study medication as metformin may have reduced clearance in this situation and therefore increased risk of adverse effects. A local guideline on the management of acute illness and prevention of acute kidney injury, such as the Kidney Health Australia (KHA) Sick Day Plan [21], may also be followed.
Study medication may be temporarily stopped for up to 12 weeks. If a participant is to recommence the study drug, the treating physician may elect to either resume the participant at the previously tolerated medication dosage or recommence up-titration as outlined above.
If the study medication is permanently discontinued, the participant will remain in the trial until the final study visit, except where the participant withdraws their consent to participate in the study.
Strategies to improve adherence to interventions {11c}
Face-to-face or remote adherence reminder sessions will be conducted by site staff when the initial study medication dispensing occurs and at each dispensing visit thereafter. Sessions will include:
-
• The importance of following study guidelines for adherence to study medication as directed
-
• Instructions on taking study medication including dose timing (particularly that the study product should be taken with the evening meal), storage and what to do in the event of a missed dose
-
• Notification of the need to return any unused study medication at each study visit to preserve the integrity of the research
-
• Reinforcement that the study medication may be metformin or placebo
-
• Importance of calling the clinic if experiencing problems possibly related to study product, including symptoms and lost pills.
Participants will also be asked about any difficulties they are experiencing taking their study medication and will be given the opportunity to ask questions.
Adherence to study medications will be monitored by tablet count at the end of the run-in phase and at each 6-monthly study visit. Participants will return any unused tablets but will not be advised that pill counting is being conducted or used as an adherence measure.
Relevant concomitant care permitted or prohibited during the trial {11d}
All participants will receive their usual CKD management as per the standard of care and local, national and international practice guidelines. This includes, but is not limited to, the management of anaemia, oedema, bone mineral disorder, cardiovascular risk factors/disease and dialysis access/transplant planning.
Blood pressure will be optimized for all participants prior to randomization. The run-in period will target a blood pressure of < 140/90 mmHg, with either up-titration of current medications or the introduction of further anti-hypertensives. This is based on the results of the HALT-PKD trials [22], which demonstrated the beneficial effect of blood pressure control in patients with ADPKD. Newly diagnosed hypertension should be treated with renin–angiotensin–aldosterone system (RAAS) blockers in the first instance unless there is a contraindication. The decision regarding individual blood pressure targets and the choice of anti-hypertensives will be at the discretion of the local treating physician, in line with relevant local and/or international guidelines.
Participants on tolvaptan will be eligible for the study providing therapy has been on a long-term stable dose, as tolvaptan results in an acute GFR decline [23]. Participants will only be eligible if they have been on the medication for at least 6 months, with a stable dose of tolvaptan for the previous 3 months. During the study, sites will be strongly encouraged to maintain the current dosage of tolvaptan, although participants may start, stop, or change dosage. Tolvaptan dose will be recorded over time. Study participants will be stratified by tolvaptan use at randomization.
Provisions for post-trial care {30}
Throughout the trial, participants will maintain usual care under their primary physician. After the trial’s completion, this standard of care will be recommended to continue.
Outcomes {12}
The primary outcome measure is the change in eGFR at 2 years from randomization. eGFR will be estimated using the CKD-EPI equation from the serum creatinine concentration collected throughout the study and analysed in local accredited laboratories [24]. Sensitivity analyses will employ the Modification of Diet in Renal Disease (MDRD) equation and cystatin C-based eGFR assessed in a central laboratory.
Secondary outcome measures are:
-
a)
Annualized slope of eGFR
-
b)
A composite outcome comprising a reduction from baseline eGFR of ≥ 30%, kidney failure (defined as an eGFR < 15 mL/min/1.73 m2) and all-cause mortality
-
c)
Reduction from baseline eGFR of ≥ 30%
-
d)
Kidney failure (defined as an eGFR < 15 mL/min/1.73 m.2)
-
e)
All-cause mortality
-
f)
The proportion of participants requiring a dosage adjustment or the introduction of a new anti-hypertensive agent during the treatment period
-
g)
Urine albumin:creatinine ratio
-
h)
Albuminuria (urine albumin:creatinine ratio) category (A1 < 3.39 mg/mmol, A2 3.39–33.9 mg/mmol, A3 > 33.9 mh/mmol) and as a continuous variable
-
i)
Health-related quality of life (QOL) scores measured using EuroQual 5 Dimensions 5 Levels (EQ-5D-5L) questionnaire [25]
-
j)
ADPKD-related pain measured using the ADPKD-PDS [26]
-
k)
Gastrointestinal symptoms measured using the Gastrointestinal Symptom Rating Scale (GSRS) [27]
-
l)
Incidence rates of gastrointestinal symptoms, lactic acidosis, deranged liver function tests, hypoglycaemia, anaemia and vitamin B12 deficiency
-
m)
Healthcare utilization (hospital admissions, non-admitted episodes of primary and specialist care and prescribed medications)
-
n)
Incremental costs and health outcomes (quality-adjusted life year (QALY) and clinically important difference in the primary outcome) of metformin therapy compared to placebo
Exploratory outcome measures will be the change in body weight and body mass index measured using height and weight measures at baseline and 2 years, and duration of interventional product usage. Other exploratory outcome measures may be formulated and proposed, but will only be incorporated with the agreement of the Global Steering Committee (GSC) and upon ethical approval, and then applied with participant consent.
Participant timeline {13}
All participants will be followed up at their respective site according to the schedule in Table 1. There are a total of 13 study visits (telephone, in person or remote). The week 12 visit will mark the end of the run-in phase and month 0 of the trial phase.
Sample size {14}
The study has been powered to detect a 20% reduction in eGFR slope due to metformin, assuming a slope of 6.9 mL/min/1.73 m2 (over 2 years; standard deviation 12; correlation 0.8) on placebo due to metformin [3, 28, 29], with 80% power at the 5% significance level. Allowing for a 10% loss to follow-up and adjustments for 5% drop-in and 5% drop-out rate, 1174 participants will be recruited.
Recruitment {15}
Participants will be recruited from kidney units that provide a comprehensive kidney specialist service in Australia, the UK, New Zealand, India, Hong Kong, South-East Asia and Europe. Pre-screening will take place at the local recruiting centres, where trained research team members will review the history of centre patients with a diagnosis of ADPKD. Patients who appear to fulfil the inclusion criteria for the study will be approached during their clinic appointments or by mail through a trial invitation letter.
The trial will also be advertised to the ADPKD community through patient-accessible websites, social media and presentations at patient information days. A Consumer advisory board will be consulted and provide input on recruitment strategies. Potential participants will undergo telephone pre-screening by members of the research team at their local recruiting centre, who may (with permission of the potential participant) contact their primary care physician and local hospital if further medical information is required to evaluate eligibility.
Potential participants who meet the eligibility criteria will be provided with a participant information sheet and the opportunity to discuss the trial and ask any questions they may have with the research team. They will then receive a follow-up phone call to determine if they would like to participate.
link