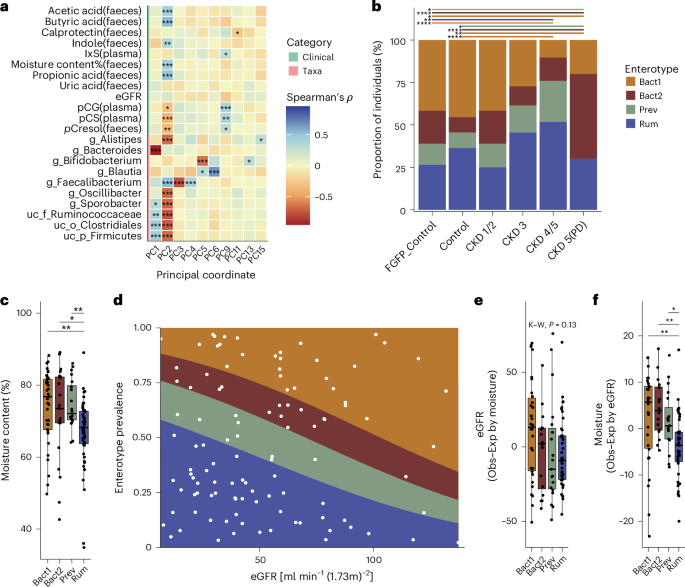
Host factors dictate gut microbiome alterations in chronic kidney disease more strongly than kidney function
Kovesdy, C. P. Epidemiology of chronic kidney disease: an update 2022. Kidney Int. Suppl. (2011) 12, 7–11 (2022).
Google Scholar
Levey, A. S. et al. Definition and classification of chronic kidney disease: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 67, 2089–2100 (2005).
Google Scholar
Levin, A. et al. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. Suppl. 3, 1–150 (2013).
Vanholder, R., Pletinck, A., Schepers, E. & Glorieux, G. Biochemical and clinical impact of organic uremic retention solutes: a comprehensive update. Toxins 10, 33 (2018).
Google Scholar
Opdebeeck, B. et al. Indoxyl sulfate and p-cresyl sulfate promote vascular calcification and associate with glucose intolerance. J. Am. Soc. Nephrol. 30, 751–766 (2019).
Google Scholar
Stadlbauer, V. et al. Structural and functional differences in gut microbiome composition in patients undergoing haemodialysis or peritoneal dialysis. Sci. Rep. 7, 15601 (2017).
Google Scholar
Margiotta, E. et al. Gut microbiota composition and frailty in elderly patients with chronic kidney disease. PLoS ONE 15, e0228530 (2020).
Google Scholar
Wang, H. et al. Perturbed gut microbiome and fecal and serum metabolomes are associated with chronic kidney disease severity. Microbiome 11, 3 (2023).
Google Scholar
Wang, X. et al. Aberrant gut microbiota alters host metabolome and impacts renal failure in humans and rodents. Gut 69, 2131–2142 (2020).
Google Scholar
Wu, I. W. et al. Integrative metagenomic and metabolomic analyses reveal severity-specific signatures of gut microbiota in chronic kidney disease. Theranostics 10, 5398–5411 (2020).
Google Scholar
Wu, I. W. et al. Gut microbiota as diagnostic tools for mirroring disease progression and circulating nephrotoxin levels in chronic kidney disease: discovery and validation study. Int. J. Biol. Sci. 16, 420–434 (2020).
Google Scholar
Yang, X. et al. Characterization of gut microbiota in patients with stage 3–4 chronic kidney disease: a retrospective cohort study. Int. Urol. Nephrol. 56, 1751–1762 (2024).
Google Scholar
Zhang, P. et al. Metagenome-wide analysis uncovers gut microbial signatures and implicates taxon-specific functions in end-stage renal disease. Genome Biol. 24, 226 (2023).
Google Scholar
Sato, N. et al. Metagenomic profiling of gut microbiome in early chronic kidney disease. Nephrol. Dial. Transplant 36, 1675–1684 (2021).
Google Scholar
Chen, T. H., Cheng, C. Y., Huang, C. K., Ho, Y. H. & Lin, J. C. Exploring the relevance between gut microbiota-metabolites profile and chronic kidney disease with distinct pathogenic factor. Microbiol. Spectr. 11, e02805–22 (2023).
Google Scholar
Peters, B. A. et al. Association of the gut microbiome with kidney function and damage in the Hispanic Community Health Study/Study of Latinos (HCHS/SOL). Gut Microbes 15, 2186685 (2023).
Google Scholar
Krukowski, H. et al. Gut microbiome studies in CKD: opportunities, pitfalls and therapeutic potential. Nat. Rev. Nephrol. 19, 87–101 (2023).
Google Scholar
Lloréns-Rico, V., Vieira-Silva, S., Gonçalves, P. J., Falony, G. & Raes, J. Benchmarking microbiome transformations favors experimental quantitative approaches to address compositionality and sampling depth biases. Nat. Commun. 12, 3562 (2021).
Google Scholar
Greenacre, M. Compositional data analysis. Annu. Rev. Stat. Appl. 8, 271–299 (2021).
Google Scholar
Nishijima, S. et al. Fecal microbial load is a major determinant of gut microbiome variation and a confounder for disease associations. Cell 188, 222–236.e15 (2025).
Google Scholar
Kanehisa, M. & Goto, S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 28, 27–30 (2000).
Google Scholar
Anatomical Therapeutic Chemical (ATC) Classification (WHO Collaborating Centre for Drug Statistics Methodology, 2024); https://www.who.int/tools/atc-ddd-toolkit/atc-classification
Amaretti, A. et al. Profiling of protein degraders in cultures of human gut microbiota. Front. Microbiol. 10, 2614 (2019).
Google Scholar
Miquel, S. et al. Faecalibacterium prausnitzii and human intestinal health. Curr. Opin. Microbiol. 16, 255–261 (2013).
Google Scholar
Arumugam, M. et al. Enterotypes of the human gut microbiome. Nature 473, 174–180 (2011).
Google Scholar
Holmes, I., Harris, K. & Quince, C. Dirichlet multinomial mixtures: generative models for microbial metagenomics. PLoS ONE 7, e30126 (2012).
Google Scholar
Vandeputte, D. et al. Quantitative microbiome profiling links gut community variation to microbial load. Nature 551, 507–511 (2017).
Google Scholar
Vieira-Silva, S. et al. Quantitative microbiome profiling disentangles inflammation- and bile duct obstruction-associated microbiota alterations across PSC/IBD diagnoses. Nat. Microbiol. 4, 1826–1831 (2019).
Google Scholar
Kalantar-Zadeh, K., Ikizler, T. A., Block, G., Avram, M. M. & Kopple, J. D. Malnutrition-inflammation complex syndrome in dialysis patients: causes and consequences. Am. J. Kidney Dis. 42, 864–881 (2003).
Google Scholar
Tito, R. Y. et al. Microbiome confounders and quantitative profiling challenge predicted microbial targets in colorectal cancer development. Nat. Med. 30, 1339–1348 (2024).
Google Scholar
Devolder, L. et al. Gut microbiome composition is associated with long-term disability worsening in multiple sclerosis. Gut Microbes 15, 2180316 (2023).
Google Scholar
Hughes, D. A. et al. Genome-wide associations of human gut microbiome variation and implications for causal inference analyses. Nat. Microbiol. 5, 1079–107 (2020).
Google Scholar
Falony, G. et al. Population-level analysis of gut microbiome variation. Science 352, 560–564 (2016).
Google Scholar
Falony, G., Vieira-Silva, S. & Raes, J. Richness and ecosystem development across faecal snapshots of the gut microbiota. Nat. Microbiol. 3, 526–528 (2018).
Google Scholar
Vandeputte, D. et al. Stool consistency is strongly associated with gut microbiota richness and composition, enterotypes and bacterial growth rates. Gut 65, 57–62 (2016).
Google Scholar
Si, J. et al. Long-term life history predicts current gut microbiome in a population-based cohort study. Nat. Aging 2, 885–895 (2022).
Google Scholar
Parthasarathy, G. et al. Relationship between microbiota of the colonic mucosa vs feces and symptoms, colonic transit, and methane production in female patients with chronic constipation. Gastroenterology 150, 367–379.e1 (2016).
Google Scholar
Cantarel, B. L. et al. The Carbohydrate-Active EnZymes database (CAZy): an expert resource for glycogenomics. Nucleic Acids Res. 37, D233–D238 (2009).
Google Scholar
Smits, S. A. et al. Seasonal cycling in the gut microbiome of the Hadza hunter-gatherers of Tanzania. Science 357, 802–806 (2017).
Google Scholar
Vieira-Silva, S. et al. Species–function relationships shape ecological properties of the human gut microbiome. Nat. Microbiol. 1, 16088 (2016).
Google Scholar
Chen, I. A. et al. The IMG/M data management and analysis system v.6.0: new tools and advanced capabilities. Nucleic Acids Res. 49, D751–D763 (2021).
Google Scholar
Almeida, A. et al. A unified catalog of 204,938 reference genomes from the human gut microbiome. Nat. Biotechnol. 39, 105–114 (2021).
Google Scholar
Procházková, N. et al. Gut physiology and environment explain variations in human gut microbiome composition and metabolism. Nat. Microbiol. 9, 3210–3225 (2024).
Google Scholar
Foreman, K. J. et al. Forecasting life expectancy, years of life lost, and all-cause and cause-specific mortality for 250 causes of death: reference and alternative scenarios for 2016–40 for 195 countries and territories. Lancet 392, 2052–2090 (2018).
Google Scholar
Vieira-Silva, S. et al. Statin therapy is associated with lower prevalence of gut microbiota dysbiosis. Nature 581, 310–315 (2020).
Google Scholar
Onal, E. M., Afsar, B., Covic, A., Vaziri, N. D. & Kanbay, M. Gut microbiota and inflammation in chronic kidney disease and their roles in the development of cardiovascular disease. Hypertens. Res. 42, 123–140 (2019).
Google Scholar
Falony, G. et al. The human microbiome in health and disease: hype or hope. Acta Clin. Belg. 74, 53–64 (2019).
Google Scholar
Gacesa, R. et al. Environmental factors shaping the gut microbiome in a Dutch population. Nature 604, 732–739 (2022).
Google Scholar
Gryp, T. et al. Isolation and quantification of uremic toxin precursor-generating gut bacteria in chronic kidney disease patients. Int. J. Mol. Sci. 21, 1986 (2020).
Google Scholar
Gryp, T. et al. Gut microbiome profiling uncovers a lower abundance of Butyricicoccus in advanced stages of chronic kidney disease. J. Pers. Med. 11, 1118 (2021).
Google Scholar
Zhang, J. et al. Dysbiosis of gut microbiota in adult idiopathic membranous nephropathy with nephrotic syndrome. Microb. Pathog. 147, 104359 (2020).
Google Scholar
Gao, B. et al. Butyrate producing microbiota are reduced in chronic kidney diseases. Sci. Rep. 11, 23530 (2021).
Google Scholar
Tanca, A. et al. Potential and active functions in the gut microbiota of a healthy human cohort. Microbiome 5, 79 (2017).
Google Scholar
Zhang, Y. et al. Gut-microbiota-derived indole sulfate promotes heart failure in chronic kidney disease. Cell Host Microbe 33, 1715–1730.e5 (2025).
Google Scholar
Beau, A. et al. A specifically designed multi-biotic reduces uremic toxin generation and improves kidney function. Gut Microbes 17, 2531202 (2025).
Google Scholar
Johnson-Martinez, J. P. et al. Aberrant bowel movement frequencies coincide with increased microbe-derived blood metabolites associated with reduced organ function. Cell Rep. Med. 5, 101646 (2024).
Google Scholar
Poesen, R. et al. The influence of dietary protein intake on mammalian tryptophan and phenolic metabolites. PLoS ONE 10, e0140820 (2015).
Google Scholar
Windey, K., De Preter, V. & Verbeke, K. Relevance of protein fermentation to gut health. Mol. Nutr. Food Res. 56, 184–196 (2012).
Google Scholar
Minnebo, Y. et al. Gut microbiota response to in vitro transit time variation is mediated by microbial growth rates, nutrient use efficiency and adaptation to in vivo transit time. Microbiome 11, 240 (2023).
Google Scholar
Derrien, M., Turroni, F., Ventura, M. & van Sinderen, D. Insights into endogenous Bifidobacterium species in the human gut microbiota during adulthood. Trends Microbiol. 30, 940–947 (2022).
Google Scholar
Li, F. et al. Cardiometabolic benefits of a non-industrialized-type diet are linked to gut microbiome modulation. Cell (2025).
Google Scholar
Ebrahim, Z. et al. The effect of ß-glucan prebiotic on kidney function, uremic toxins and gut microbiome in stage 3 to 5 chronic kidney disease (CKD) predialysis participants: a randomized controlled trial. Nutrients 14, 805 (2022); erratum 17, 3054 (2025).
Sinha, A. K. et al. Dietary fibre directs microbial tryptophan metabolism via metabolic interactions in the gut microbiota. Nat. Microbiol. 9, 1964–1978 (2024).
Google Scholar
Carrero, J. J. et al. Plant-based diets to manage the risks and complications of chronic kidney disease. Nat. Rev. Nephrol. 16, 525–542 (2020).
Google Scholar
Babich, J. S., Kalantar-Zadeh, K. & Joshi, S. Taking the kale out of hyperkalemia: plant foods and serum potassium in patients with kidney disease. J. Ren. Nutr. 32, 641–649 (2022).
Google Scholar
Fromentin, S. et al. Microbiome and metabolome features of the cardiometabolic disease spectrum. Nat. Med. 28, 303–314 (2022).
Google Scholar
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014).
Google Scholar
Kim, K., Cho, W. H., Hwang, S. D., Lee, S. W. & Song, J. H. Association between constipation and incident chronic kidney disease in the UK Biobank study. Sci. Rep. 14, 32106 (2024).
Google Scholar
Gryp, T. et al. Gut microbiota generation of protein-bound uremic toxins and related metabolites is not altered at different stages of chronic kidney disease. Kidney Int. 97, 1230–1242 (2020).
Google Scholar
National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am. J. Kidney Dis. 39, S1–S266 (2002).
Gansevoort, R. T. et al. Recommendations for the use of tolvaptan in autosomal dominant polycystic kidney disease: a position statement on behalf of the ERA-EDTA Working Groups on Inherited Kidney Disorders and European Renal Best Practice. Nephrol. Dial. Transplant. 31, 337–348 (2016).
Google Scholar
Steenbeke, M. et al. Gut microbiota and their derived metabolites, a search for potential targets to limit accumulation of protein-bound uremic toxins in chronic kidney disease. Toxins 13, 809 (2021).
Google Scholar
Wang, Q., Garrity, G. M., Tiedje, J. M. & Cole, J. R. Naïve Bayesian Classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 73, 5261–5267 (2007).
Google Scholar
Oksanen, J. et al. vegan: Community Ecology Package v.2.6-4 (CRAN, 2022).
Ma, S. et al. Population structure discovery in meta-analyzed microbial communities and inflammatory bowel disease using MMUPHin. Genome Biol. 23, 208 (2022).
Google Scholar
Kuhn, M. Building predictive models in R Using the caret package. J. Stat. Softw. 28, 1–26 (2008).
Google Scholar
Ho, D., Imai, K., King, G. & Stuart, E. A. MatchIt: nonparametric preprocessing for parametric causal inference. J. Stat. Softw. 42, 1–28 (2011).
Google Scholar
Bushner, B. BBMap: BBMap short read aligner, and other bioinformatic tools. v.38.51 sourceforge.net/projects/bbmap/ (2015).
Nurk, S., Meleshko, D., Korobeynikov, A. & Pevzner, P. A. metaSPAdes: a new versatile metagenomic assembler. Genome Res. 27, 824–834 (2017).
Google Scholar
Kang, D. D., Froula, J., Egan, R. & Wang, Z. MetaBAT, an efficient tool for accurately reconstructing single genomes from complex microbial communities. PeerJ 3, e1165 (2015).
Google Scholar
Ruscheweyh, H. J. et al. Cultivation-independent genomes greatly expand taxonomic-profiling capabilities of mOTUs across various environments. Microbiome 10, 212 (2022).
Google Scholar
Hyatt, D. et al. Prodigal: prokaryotic gene recognition and translation initiation site identification. BMC Bioinformatics 11, 119 (2010).
Google Scholar
Fu, L., Niu, B., Zhu, Z., Wu, S. & Li, W. CD-HIT: accelerated for clustering the next-generation sequencing data. Bioinformatics 28, 3150–3152 (2012).
Google Scholar
Cantalapiedra, C. P., Hernandez-Plaza, A., Letunic, I., Bork, P. & Huerta-Cepas, J. eggNOG-mapper v2: functional annotation, orthology assignments, and domain prediction at the metagenomic scale. Mol. Biol. Evol. 38, 5825–5829 (2021).
Google Scholar
Woodcroft, B. J. CoverM: read alignment statistics for metagenomics. v.0.6.1 (2021).
Gregory, A. C. et al. MetaPop: a pipeline for macro- and microdiversity analyses and visualization of microbial and viral metagenome-derived populations. Microbiome 10, 49 (2022).
Google Scholar
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Google Scholar
Danecek, P. et al. Twelve years of SAMtools and BCFtools. Gigascience 10, giab008 (2021).
Google Scholar
Valles-Colomer, M. et al. The neuroactive potential of the human gut microbiota in quality of life and depression. Nat. Microbiol. 4, 623–632 (2019).
Google Scholar
Buades Fuster, J. M., Sanchis Cortes, P., Perello Bestard, J. & Grases Freixedas, F. Plant phosphates, phytate and pathological calcifications in chronic kidney disease. Nefrologia 37, 20–28 (2017).
Google Scholar
Caspi, R. et al. The MetaCyc database of metabolic pathways and enzymes—a 2019 update. Nucleic Acids Res. 48, D445–D453 (2020).
Google Scholar
Darzi, Y., Falony, G., Vieira-Silva, S. & Raes, J. Towards biome-specific analysis of meta-omics data. ISME J. 10, 1025–1028 (2016).
Google Scholar
Davison, A. C. & Hinkley, D. V. Bootstrap Methods and Their Application (Cambridge Univ. Press, 1997).
Canty, A. & Ripley, B. D. boot: bootstrap R (S-Plus) functions. R version v.1.3-28.1 (2022).
Champely, S. pwr: basic functions for power analysis. v.1.3-0 (2020).
Ferdous, T. et al. The rise to power of the microbiome: power and sample size calculation for microbiome studies. Mucosal Immunol. 15, 1060–1070 (2022).
Google Scholar
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2022).
Yoshida, K. et al. tableone: create ‘Table 1’ to describe baseline characteristics with or without propensity score weights. v.0.13.2 (2022).
Ogle, D. H., Doll, J. C., Wheeler, A. P. & Dinno, A. FSA: simple fisheries stock assessment method. R package version 0.9.5 (2023).
McMurdie, P. J. & Holmes, S. phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data v.1.42.0. PLoS ONE 8, e61217 (2013).
Kassambara, A. rstatix: pipe-friendly framework for basic statistical tests. R version v.0.7.2 (2023).
Martinez Arbizu, P. pairwiseAdonis: pairwise multilevel comparison using adonis. R version v.0.4.1 (2020).
Lathi, L. & Shetty, S. microbiome R package. Bioconductor (2017).
Proost, S., Vieira-Silva, S. & Raes, J. LoreplotR: logistic regression plots in R. v.0.2.1 (2023).
Krukowski, H. raeslab/ckd-ghent: CKD-ghent. Zenodo (2026).
link