Healthy dietary patterns and the incidence of chronic kidney disease: results from a prospective cohort study | BMC Public Health

Study population
This prospective study draws on data from the ongoing UK Biobank cohort (Application number: 51671; approved in August 2019), which includes over 500,000 participants aged 40–69 years, recruited across various regions of England, Wales, and Scotland between 2006 and 2010. Detailed information on the study design and assessment protocols of the UK Biobank cohort has been previously published [26]. Ethical approval was granted by the National Information Governance Board for Health and Social Care in England and Wales, and the Community Health Index Advisory Group in Scotland. Written informed consent was obtained from all participants prior to data collection.
Participants with missing 24-hour recall data or implausible daily energy intake (total energy intake < 2508 or > 14630 kj/day in women, and < 3344 or > 17556 kj/day in men) [27] were excluded. Ultimately, 210,950 participants were included in the analysis. The baseline was defined as the date of the first 24-hour dietary recall assessment in 2011, continuing until the earliest of the following events: the first CKD diagnosis, death, or the end of the follow-up period. Participants who had died prior to the baseline or had a history of CKD (International Classification of Diseases, Tenth Revision [ICD-10] code N18) or CKD-related conditions (ICD-10 codes I13, N02, N03, N04, N05, N06, N07, N08, and N15) before the baseline (n = 3,653) were excluded. Additionally, participants lost to follow-up (n = 29) were also excluded. Consequently, the final analysis included 207,268 participants (92,884 men and 114,384 women).
Assessment of outcomes
The definition of CKD events in this study included the initial diagnosis of CKD (ICD-10 code N18), an estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m² at any point during follow-up, or a urine albumin-creatinine ratio ≥ 30 mg/g at any time during the follow-up period.
Assessment of the dietary scores
At the recruitment assessment center, participants were invited by email to complete an online 24-hour dietary recall assessment. This assessment collected data on the consumption of 206 food items and 32 beverage types over the preceding 24 h, along with estimates of energy and nutrient intake. Participants were also required to report portion sizes for each food item, specifying the number of servings consumed. Standard portion sizes were provided for most items, including one apple, one sausage, one rasher of bacon, or one slice of ham. For foods like pasta or rice, a “serving” was defined, and participants were asked to indicate the number of servings consumed.
The associations between four distinct healthy dietary patterns and CKD risk were analyzed, including the hPDI, HEI-2015, MED Score, and AMED Score. Detailed descriptions of the components and scoring criteria for each dietary score were provided in Supplemental Tables S1-S4. The hPDI included 18 food groups, categorized into quintiles of consumption, with each quintile assigned a score from 1 to 5, either positive or reverse, yielding a total score ranging from 18 to 90. The HEI-2015 consisted of 13 components, with a total score range of 0 to 100. Both the MED Score and AMED Score encompassed nine components, each with a total score range of 0 to 9. Higher scores indicated greater adherence to the respective dietary patterns.
CKD prediction model
The prediction model developed by Nelson et al. was used to estimate the risk of CKD incidence [28]. Given that variables such as albuminuria and HbA1c frequently differ based on diabetes status, separate models were constructed for participants with and without diabetes. The primary model for predicting 5-year risk of reduced eGFR included demographic factors (age, sex, and race/ethnicity), eGFR (modeled using linear splines with a knot at 90 mL/min/1.73 m2), history of cardiovascular disease, smoking, hypertension, body mass index (BMI), and albuminuria. For diabetic participants, the model further accounted for diabetes medications, HbA1c levels, and their interaction. The prediction equation was provided in Supplemental Table S9, with stratification into quartiles for analysis.
Covariates
Covariates were obtained through participant interviews conducted during the recruitment phase. These included age (years), sex (female or male), assessment center, ethnic background (white or non-white), index of multiple deprivation (IMD), BMI category (low, normal, overweight, or obese), smoking status (never, former, or current), alcohol consumption (special occasions/never, 1–3 times/month, 1–4 times/week, or daily/almost daily), physical activity (metabolic equivalent hours/week), sleep duration (< 8 h, 8 h, 8–9 h, or > 9 h), history of hypertension (yes or no), history of diabetes (yes or no), general health status (self-reported long-standing illness), use of dietary supplements (no, yes, unknown, or prefer not to answer), and drug use (yes or no), including nonsteroidal anti-inflammatory drugs (NSAIDs), angiotensin-converting enzyme inhibitors (ACEIs), angiotensin receptor blockers (ARBs), and beta-blockers.
Statistical analysis
Descriptive statistics were used to assess the baseline characteristics of participants across different dietary patterns. Person-years were calculated from the date of the initial 24-hour dietary recall to the earliest of the following events: CKD diagnosis, death, or end of the follow-up period. Normally distributed continuous variables were presented as mean (SD), and categorical variables as number (percentages). Cox proportional hazards models were applied to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for the association between healthy dietary scores and CKD risk. Multivariable logistic regression models were employed to examine the relationship between incident CKD and each quantile of the hPDI, HEI-2015, MED Score, and AMED Score, calculating HRs and corresponding 95% CIs. The first quintile (Q1) for the hPDI and HEI-2015, and the lowest score range (0–2) for the MED Score and AMED Score were designated as reference categories. The basic model adjusted for age, sex, and assessment center, while the multivariable model further accounted for ethnic background, IMD, BMI, smoking and drinking status, physical activity, sleep duration, use of dietary supplements (vitamins and minerals), drug use (NSAIDs, ACEIs, ARBs, and beta-blockers), history of hypertension and diabetes, general health status (self-reported long-standing illness) and total energy intake. CKD risk per standard deviation increase in the dietary scores was estimated by treating the scores as continuous variables. To reduce the impact of reverse causality, one-year lag analyses were performed (Fig. 1).
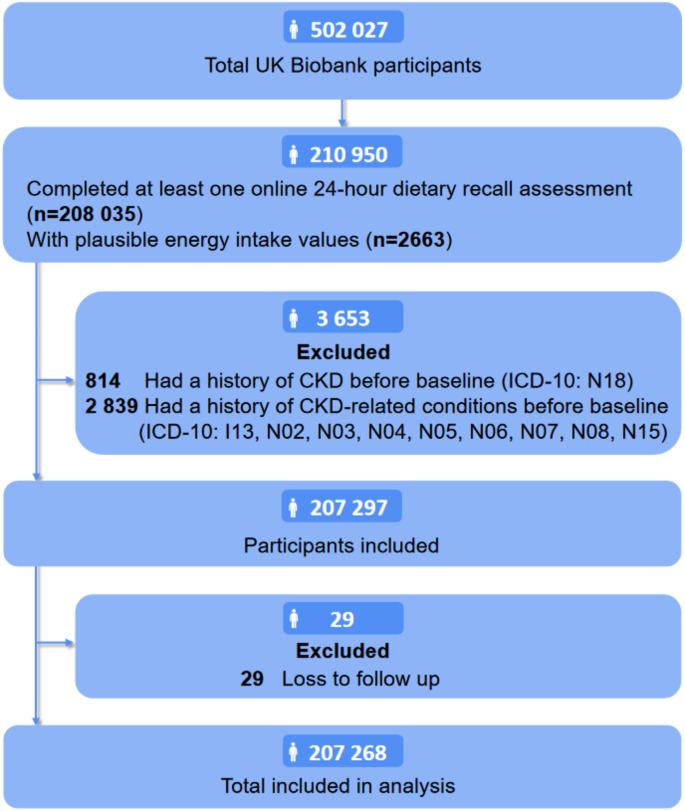
Flow of participants through study
Sensitivity analyses were conducted to assess the robustness of the study. To mitigate reverse causation from pre-existing conditions, 2-year and 4-year lag analyses were implemented. Participants diagnosed with diabetes mellitus or hypertension at baseline were excluded prior to analysis. Sensitivity analysis was conducted before(Model 1) and after (Model 2) including total energy intake as a correction variable. Subgroup analyses were performed, and statistical interactions evaluated based on potential risk factors, including sex, baseline age (2006), BMI, physical activity, smoking status, drinking habits, and health status. Two-sided statistical tests were used, with p < 0.05 considered statistically significant. An interaction term was incorporated into the regression model to examine the relationship between dietary scores and various covariates. The analysis adhered to the STROBE cohort reporting guidelines [29].
link