Digital health technology use in clinical trials of rare diseases: a systematic review
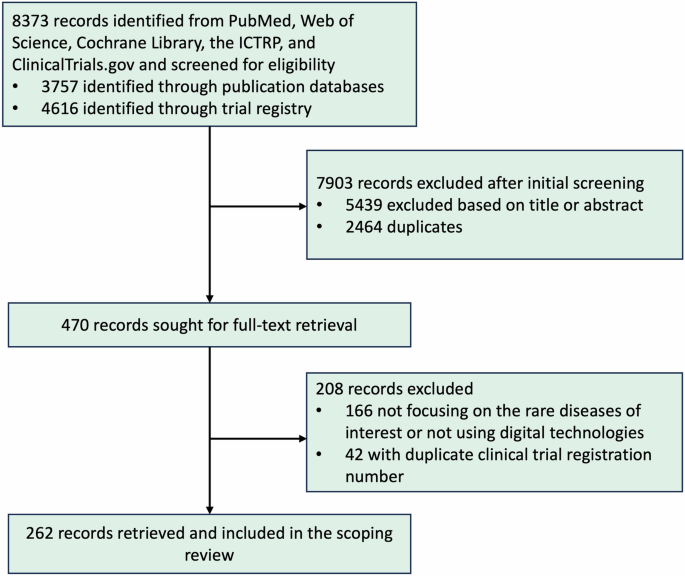
Over the past few decades, substantial global efforts have been undertaken to advance therapeutic development for rare diseases through supportive regulatory policies and economic incentives24,25. In the US, landmark initiatives such as the Orphan Drug Act (1983)26 and the Accelerating Rare disease Cures program (2022) have promoted this mission by driving scientific innovation, regulatory flexibility and stakeholder engagement27. Likewise, the European Medicines Agency (EMA) has introduced a suite of incentives, including market exclusivity, fee reductions, and scientific advice, to stimulate orphan drug development. In China, supportive policies like accelerated approval pathways for urgently needed orphan drugs (UNOD) and Care for Rare Diseases Encouragement (CARE) plan28, have been implemented to improve access to therapies for rare conditions. Despite these advances, approved treatments remain available for only 5~7% of the estimated 7000 to 10,000 rare diseases worldwide29. Clinical trials represent the most resource-intensive and failure-prone phase of drug R&D—and are even more complex in the context of rare diseases (Table 1). Small, geographically dispersed patient populations and high phenotypic variability often lead to slow recruitment, limited statistical power, and extended trial durations30,31,32,33,34. Addressing these challenges is critical to advancing robust and inclusive therapeutic research for underserved populations.
Our study highlights a significant upward trend in the deployment of DHTs in interventional trials for selected rare diseases, particularly since 2020 (Fig. 3). To capture the most recent developments, we conducted a supplementary search extending through the end of 2024, identifying nearly 60 additional DHT-enabled rare disease trials initiated in that year alone. Returning to our primary dataset, we found that in certain conditions such as CF and FTD, over 20% of recent trials incorporated digital components—a proportion higher than the reported 11% adoption rate in trials for common neurological disorders in 202035. These findings raise important questions about what makes digital technologies increasingly valuable in the rare disease trial landscape, and what benefits they offer beyond logistical convenience.
DHTs enhance trial accessibility by enabling remote participation, thereby mitigating recruitment delays caused by small, scattered populations. For instance, telemedicine platforms such as Amwell and Teladoc Health enable remote consultation with specialists, and eConsent platforms like Medidata eConsent and REDCap streamline informed consent processes and reduce in-person requirements. Beyond accessibility, DHTs also enhance operational agility by simplifying workflows and providing more patient-friendly options. Wearable biosensors (e.g., ActiGraph, Biofourmis) and mobile health (mHealth) applications (e.g., Apple ResearchKit, Google Fit) support real-time home-based monitoring of vital signs, medication adherence, and disease progression. Electronic clinical outcome assessment (eCOA) platforms, such as Medable and Clinical Ink, enable patients to report symptoms and QoL metrics in a timely and structured manner.
In addition to improving accessibility and agility, DHTs also hold promising potential to improve therapeutic outcomes in rare diseases. Our analysis of controlled studies with available results highlights the feasibility and benefits of various digital therapeutic and long-term care interventions (Supplementary Data 2), particularly in symptom relief and QoL improvements. For example, an automated text message based intervention successfully increased physical activity in patients with PAH36 (p = 0.02). A home-based programme using active video games effectively improved exercise capacity, muscular strength and short-term QoL in children and adolescents with CF (p < 0.05)22. However, conducting a meta-analysis across studies remains challenging due to heterogeneity in disease characteristics, clinical outcomes, and evaluation metrics. Nonetheless, the available evidence underscores the clinical relevance and future promise of DHTs in supporting care for rare disease populations.
Current applications of DHTs are predominantly concentrated in four domains: digital treatment, data monitoring and collection, long-term healthcare and remote follow-up (Table 2). Despite the transformative potential of DHTs, their effective implementation requires alignment with the specific clinical contexts and needs of each disease. This is reflected in our findings, which reveal marked variation in DHT adoption across the selected diseases. For instance, CF trials exhibited the highest level of digital utilization across multiple domains, owing to its complex and chronic nature, which necessitates continuous monitoring, long-term care management, and proactive intervention to maintain lung function and prevent exacerbations37. ALS studies showed substantial use of data monitoring technologies, reflecting the critical need to track rapid disease progression and manage associated symptoms. These patterns underscore the importance of tailoring DHT strategies to disease-specific characteristics to maximize their clinical relevance and impact.
Across the entire rare disease trial continuum, DHTs hold promise to support patient-centric approaches—from trial design to patient recruitment and retention, and to data monitoring, outcome assessment, and long-term healthcare (Fig. 4). However, as our analysis primarily draws from trial registries and published literature, it may not fully capture the breadth or depth of DHT integration across all stages of clinical research. Additionally, given the existence of over 10,000 rare diseases, and especially the scarcity of research on ultra-rare conditions, capturing the full landscape remains inherently challenging. To complement this view, we conducted supplementary searches on Google Scholar and grey literature, aiming to provide a broader, though non-exhaustive, picture of DHT applications beyond formal trial registries and publications.
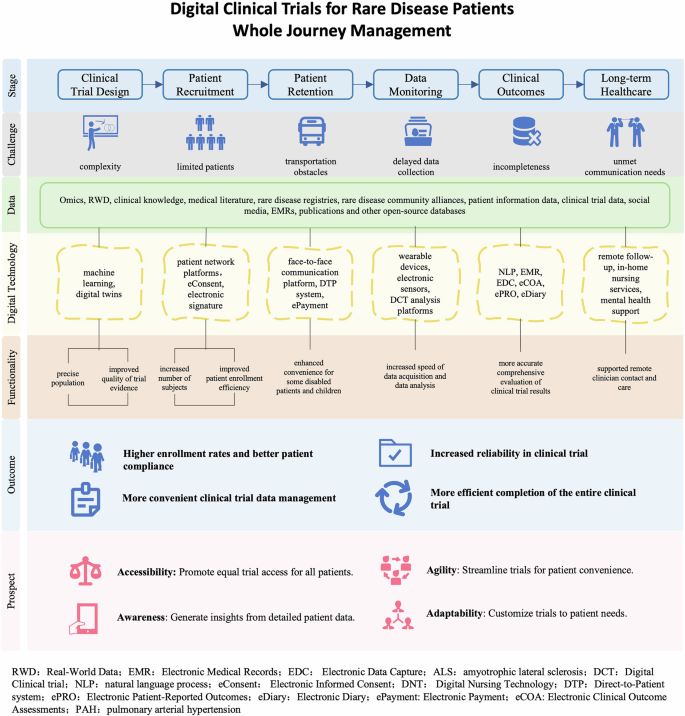
This framework illustrates how DHTs can address specific challenges throughout the clinical trial continuum—from trial design, patient recruitment, retention, and monitoring, to outcome evaluation and long-term healthcare. It maps available data types, relevant digital tools, and their functionalities, highlighting how DHTs enhance patient access, operational efficiency, and trial robustness. The “4A” prospect—Accessibility, Agility, Awareness, and Adaptability—summarizes the broader value of DHTs for rare disease research.
During the trial design phase, digital approaches such as machine learning algorithms and digital twin models enable precise cohort selection and the generation of predictive models to optimize sample sizes—an essential feature for rare diseases with limited patient pools. Platforms like TwinRCTsTM38, Clinerion39, and Deep 6 AI40 have been leveraged to support more data-driven, patient-tailored trial designs41. In the recruitment phase, digital platforms broaden patient outreach and improve enrollment efficiency7. Patient network systems such as the US National Institutes of Health’s Rare Diseases Clinical Research Network42 and the National Rare Diseases Registry System of China43 facilitate access to geographically dispersed populations. To support patient retention, direct-to-patient (DTP) drug delivery systems and digital communication platforms ease logistics and improve compliance, particularly for patients with continuous care needs or with mobility limitations. Digital treatment modalities, such as mental health therapies and game-based interventions, offer the psychological support and engagement particularly suited to pediatric rare disease patients44,45. Home-based physiological monitoring using wearable devices and mobile apps is also becoming more widespread46. Beyond data acquisition, platforms such as Medidata cloud platform47, Veeva Systems48 and Oracle Health Sciences49 provide integrated infrastructure for real-time data processing, disease monitoring and outcome assessment. In the context of long-term healthcare, DHTs help bridge persistent communication gaps between patients and healthcare providers. They support remote consultations, in-home nursing services and online training programs, collectively contributing to improved QoL for patients50. Together, these tools illustrate how DHTs can be strategically applied across trial stages to address challenges specific to rare disease populations.
Looking ahead, to better navigate and consolidate the complex landscape of DHT applications in rare disease research, we propose an integrated framework (Fig. 4) that maps digital tools onto six key stages of the trial process: from trial design to long-term healthcare. This framework outlines stage-specific challenges, identifies applicable data types and digital technologies, highlights how DHTs can improve trial efficiency, data quality, and patient engagement. Building on this structure, we recommend that future DHT deployment can be guided by four foundational principles—Accessibility, Agility, Awareness, and Adaptability (“4A”). These principles provide a strategic lens to ensure more inclusive, efficient, and patient-centered research.
Accessibility ensures that patients with rare diseases have equitable opportunities to engage in research through remote access and decentralized trial model30,51. Agility emphasizes streamlined trial operations to reduce logistical burdens, enabling seamlessly participation through wearable devices, home-based monitoring systems, and virtual consultations—particularly beneficial for those managing chronic and progressive conditions. Awareness underscores the value of real-world data, and patient-reported outcomes in deepening understanding of rare disease biology, progression, and patient needs—filling knowledge gaps that often hinder therapeutic progress. Adaptability reflects the need to tailor studies to the evolving needs of rare disease populations, where the focus extends beyond survival to improving QoL and ensuring patients can live with greater dignity.
As technology continues to evolve and regulatory frameworks adapt, the adoption of DHTs in rare disease clinical trials is poised to expand further. Numerous emerging DHTs, such as next-generation wearable devices, large language models52, and digital twins53, are expected to revolutionize clinical trial paradigm54. For instance, medical digital twins—computational models that replicates human biology relevant to a compound or intervention—are gaining traction in medical research55. These advancements herald a shift towards virtual clinical trials56, which provide an efficient and scalable alternative for rare diseases with limited patient pools. Meanwhile, large language model (LLM) technologies are reshaping conventional clinical development processes, facilitating patient matching57,58, protocol design59, and early prediction of trial outcomes60.
However, despite their transformative potential, DHTs in clinical trials still face multifaceted challenges61. Firstly, safeguarding patient data remains paramount. This requires robust data governance frameworks, end-to-end encryption, and secure, interoperable data hubs62 to ensure compliance with The Health Insurance Portability and Accountability Act of 1996 (HIPAA), The General Data Protection Regulation (GDPR), and other regional regulations. Secondly, ensuring data quality and operational efficiency requires the integration of advanced management systems with real-time monitoring, AI-driven analytics, and standardized data formats. However, differences in device calibration, data transmission, and software compatibility pose significant challenges for seamless data integration. Adopting standards such as Fast Healthcare Interoperability Resources (FHIR) and Clinical Data Interchange Standards Consortium (CDISC), along with automated validation tools, can facilitate data harmonization and interoperability across platforms. Third, integrating AI into clinical trials raises ethical concerns, including algorithmic bias, lack of explainability of decision-making, and risks of hallucinations or misdiagnoses that could negatively impact patient outcomes. These issues necessitate the development of dedicated ethical and regulatory frameworks. Fourth, ensuring digital equity remains a critical concern30. Unequal access to digital devices and reliable internet connectivity, particularly in low- and middle-income countries (LMICs), impedes participation in digital trials. Socioeconomic disparities, limited digital literacy, and language barriers further exacerbate exclusion. To address the language gap for First Nations people, the DHT-enabled Lyfe Languages platform, which initially launched a rare disease module, plans to expand its translations and educational resources into additional domains such as clinical trials and treatment information. Beyond infrastructure limitations, certain patient populations, such as the elderly and individuals with physical or cognitive impairments, may face difficulties in interacting with wearable devices and telemedicine platforms. Addressing these disparities requires efforts to enhance accessibility, strengthen infrastructure, and design inclusive digital solutions. Ensuring equitable participation is essential to maximizing the impact of DHTs in clinical research63,64.
This study has several limitations. First, the historical absence of a globally unified definition of rare diseases constrained our study scope to the top ten rare diseases with the highest R&D activity, which may not fully represent the broader spectrum of rare disease research. This approach inevitably excludes some less-studied conditions, particularly those with very small patient populations or those that have only recently been the focus of pharmaceutical interest, but it nonetheless captures the lion’s share of DHT investment and innovation in the rare disease space. Indeed, as of 2024, the 10th-ranked disease, GVHD, had only seven DHT-enabled trials, suggesting that this focus is sufficient to capture key trends. Second, the application of DHTs is often underreported or undocumented, making it difficult to assess their full usage. To mitigate this, we conducted supplementary searches and developed an end-to-end “4A” application framework to provide a more comprehensive perspective.
In conclusion, this study reveals a growing trend in the integration of DHTs into patient-centric clinical trials for rare diseases, particularly in digital therapeutics, data monitoring and long-term care management. By enabling more inclusive, efficient, and tailored trial designs, these technologies pave the way for a paradigm shift in rare disease research. Beyond mapping current use cases, we propose a forward-looking framework centered on four key principles—Accessibility, Agility, Awareness, and Adaptability—to guide the meaningful application of DHTs. As technological innovations and supportive policies evolve, DHTs hold immense potential to redefine clinical research, creating a future where rare disease patients have equitable access to effective therapies and improved health outcomes. We call on researchers and policymakers to embrace the 4A framework and strengthen cross‑sector collaboration, ensuring that DHTs reach their full potential in improving rare disease research and patient care.
link