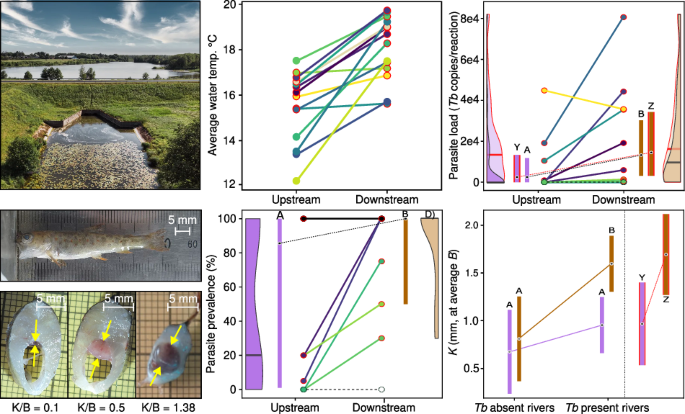
Dams threaten salmonids by triggering temperature-dependent proliferative kidney disease
Belletti, B. et al. More than one million barriers fragment Europe’s rivers. Nature 588, 436–441 (2020).
Grill, G. et al. Mapping the world’s free-flowing rivers. Nature 569, 215–221 (2019).
Zarfl, C. et al. Future large hydropower dams impact global freshwater megafauna. Sci. Rep. 9, 18531 (2019).
Su, G. et al. Human impacts on global freshwater fish biodiversity. Science 371, 835–838 (2021).
Auffray, M., Senécal, J.-F., Turgeon, K., St-Hilaire, A. & Maheu, A. Reservoirs regulated by small dams have a similar warming effect than lakes on the summer thermal regime of streams. Sci. Total Environ. 869, 161445 (2023).
Bednarek, A. Undamming rivers: a review of the ecological impacts of dam removal. Environ. Manag. 27, 803–814 (2001).
Barbarossa, V. et al. Impacts of current and future large dams on the geographic range connectivity of freshwater fish worldwide. Proc. Natl. Acad. Sci. USA 117, 3648–3655 (2020).
Palmer, M. A. et al. Climate change and the world’s river basins: anticipating management options. Front. Ecol. Environ. 6, 81–89 (2008).
Deemer, B. R. et al. Greenhouse gas emissions from reservoir water surfaces: a new global synthesis. BioScience 66, 949–964 (2016).
Olden, J. D. & Naiman, R. J. Incorporating thermal regimes into environmental flows assessments: modifying dam operations to restore freshwater ecosystem integrity. Freshw. Biol. 55, 86–107 (2010).
Sinokrot, B. A., Stefan, H. G., McCormick, J. H. & Eaton, J. G. Modeling of climate change effects on stream temperatures and fish habitats below dams and near groundwater inputs. Clim. Change 30, 181–200 (1995).
Zaidel, P. A. et al. Impacts of small dams on stream temperature. Ecol. Indic. 120, 106878 (2021).
Pereira, H. R., Gomes, L. F., Barbosa, H. D. & Agostinho, A. A. Research on dams and fishes: determinants, directions, and gaps in the world scientific production. Hydrobiologia 847, 579–592 (2020).
Marcos-López, M., Gale, P., Oidtmann, B. C. & Peeler, E. J. Assessing the impact of climate change on disease emergence in freshwater fish in the United Kingdom. Transbound. Emerg. Dis. 57, 293–304 (2010).
Bartholomew, J. L. et al. Deconstructing dams and disease: predictions for salmon disease risk following Klamath River dam removals. Front. Ecol. Evol. 11 (2023).
Ros, A., Schmidt-Posthaus, H. & Brinker, A. Mitigating human impacts including climate change on proliferative kidney disease in salmonids of running waters. J. Fish Dis. 45, 497–521 (2022).
Hartikainen, H., Johnes, P., Moncrieff, C. & Okamura, B. Bryozoan populations reflect nutrient enrichment and productivity gradients in rivers. Freshw. Biol 54, 2320–2334 (2009).
Bettge, K., Segner, H., Burki, R., Schmidt-Posthaus, H. & Wahli, T. Proliferative kidney disease (PKD) of rainbow trout: temperature- and time-related changes of Tetracapsuloides bryosalmonae DNA in the kidney. Parasitology 136, 615–625 (2009).
Okamura, B., Hartikainen, H., Schmidt-Posthaus, H. & Wahli, T. Life cycle complexity, environmental change and the emerging status of salmonid proliferative kidney disease. Freshw. Biol. 56, 735–753 (2011).
Waldner, K., Borkovec, M., Borgwardt, F., Unfer, G. & El-Matbouli, M. Effect of water temperature on the morbidity of Tetracapsuloides bryosalmonae (Myxozoa) to brown trout (Salmo trutta) under laboratory conditions. J. Fish Dis. 44, 1005–1013 (2021).
Ros, A. et al. Current and projected impacts of the parasite Tetracapsuloides bryosalmonae (causative to proliferative kidney disease) on Central European salmonid populations under predicted climate change. Freshw. Biol 66, 1182–1199 (2021).
Isaak, D. J. et al. Global warming of salmon and trout rivers in the northwestern U.S.: road to ruin or path through purgatory?. Trans. Am. Fish. Soc. 147, 566–587 (2018).
Donadi, S. et al. Contrasting long-term trends in juvenile abundance of a widespread cold-water salmonid along a latitudinal gradient: effects of climate, stream size and migration strategy. Ecography,2023, e06522 (2023).
Hedrick, R., MacConnell, E. & De Kinkelin, P. Proliferative kidney disease of salmonid fish. Annu. Rev. Fish Dis. 3, 277–290 (1993).
Zarri, L. J., Palkovacs, E. P., Post, D. M., Therkildsen, N. O. & Flecker, A. S. The evolutionary consequences of dams and other barriers for riverine fishes. BioScience 72, 431–448 (2022).
Lessard, J. L. & Hayes, D. B. Effects of elevated water temperature on fish and macroinvertebrate communities below small dams. River Res. Appl. 19, 721–732 (2003).
Schmidt-Posthaus, H., Hirschi, R. & Schneider, E. Proliferative kidney disease in brown trout: infection level, pathology and mortality under field conditions. Dis. Aquat. Org. 114, 139–146 (2015).
Strepparava, N. et al. Temperature-related parasite infection dynamics: the case of proliferative kidney disease of brown trout. Parasitology 145, 281–291 (2018).
Økland, J. & Økland, K. A. Freshwater bryozoans (Bryozoa) of Norway V: review and comparative discussion of the distribution and ecology of the 10 species recorded. Hydrobiologia 534, 31–55 (2005).
Engel, F. et al. Phytoplankton gross primary production increases along cascading impoundments in a temperate, low-discharge river: Insights from high frequency water quality monitoring. Sci. Rep. 9, 6701 (2019).
Davis, J. M., Rosemond, A. D., Eggert, S. L., Cross, W. F. & Wallace, J. B. Long-term nutrient enrichment decouples predator and prey production. Proc. Natl. Acad. Sci. USA 107, 121–126 (2009).
Wood, T. S. Digestion and nutrition in freshwater bryozoans (Phylactolaemata). Invertebr. Biol. 140, e12314 (2021).
Duval, E. et al. When does a parasite become a disease? eDNA unravels complex host-pathogen dynamics across environmental stress gradients in wild salmonid populations. Sci. Total Environ. 946, 174367 (2024).
Carraro, L. et al. Integrated field, laboratory, and theoretical study of PKD spread in a Swiss prealpine river. Proc. Natl. Acad. Sci. USA 114, 11992–11997 (2017).
Ahmad, F. et al. The strength and form of natural selection on transcript abundance in the wild. Mol. Ecol. 30, 2724–2737 (2020).
Bailey, C., Segner, H., Casanova-Nakayama, A. & Wahli, T. Who needs the hotspot? The effect of temperature on the fish host immune response to Tetracapsuloides bryosalmonae the causative agent of proliferative kidney disease. Fish Shellfish Immunol. 63, 424–437 (2017).
Bruneaux, M. et al. Parasite infection and decreased thermal tolerance: impact of proliferative kidney disease on a wild salmonid fish in the context of climate change. Funct. Ecol. 31, 216–226 (2016).
Nagrodski, A., Raby, G. D., Hasler, C. T., Taylor, M. K. & Cooke, S. J. Fish stranding in freshwater systems: sources, consequences, and mitigation. J. Environ. Manag. 103, 133–141 (2012).
Huntingford, F. A. et al. How juvenile Atlantic salmon, Salmo salar L., respond to falling water levels: experiments in an artificial stream. Fish. Manage. Ecol. 6, 357–364 (1999).
Jonsson, B. & Jonsson, N. A review of the likely effects of climate change on anadromous Atlantic salmon Salmo salar and brown trout Salmo trutta, with particular reference to water temperature and flow. J. Fish Biol. 75, 2381–2447 (2009).
Schisler, G. J., Walker, P. G., Chittum, L. A. & Bergersen, E. P. Gill ectoparasites of juvenile rainbow trout and brown trout in the upper Colorado River. J. Aquat. Anim. Health 11, 170–174 (1999).
Marcogliese, D. J. The impact of climate change on the parasites and infectious diseases of aquatic animals. Rev. Sci. Tech. 27, 467–484 (2008).
Anderson, R. M. & May, R. M. The invasion, persistence and spread of infectious diseases within animal and plant communities. Philos. Trans. R. Soc. Lond. B Biol. Sci. 314, 533–570 (1986).
Schmidt-Posthaus, H. et al. The role of migration barriers for dispersion of Proliferative Kidney Disease—Balance between disease emergence and habitat connectivity. PLoS ONE 16, e0247482 (2021).
Bellmore, J. R. et al. Conceptualizing ecological responses to dam removal: if you remove it, what’s to come?. BioScience 69, 26–39 (2018).
Dolan, E. J., Soto, I., Dick, J. T. A., He, F., Cuthbert, R. N. Riverine barrier removals could proliferate biological invasions. Glob. Change Biol. 31, e70093 (2025).
Altizer, S., Bartel, R. & Han, B. A. Animal migration and infectious disease risk. Science 331, 296–302 (2011).
Oexle, S., Ros, A. & Brinker, A. Evidence that wild salmonids seek cool water refuges to reduce parasite virulence: the proliferative kidney disease case. Freshw. Biol. 70, 7 (2025).
Kalny, G. et al. The influence of riparian vegetation shading on water temperature during low flow conditions in a medium sized river. Knowl. Manag. Aquat. Ecosyst. 418, 5 (2017).
Wohl, E., Lane, S. N. & Wilcox, A. C. The science and practice of river restoration. Water Resour. Res. 51, 5974–5997 (2015).
Myrstener, M., Greiser, C. & Kuglerová, L. Downstream temperature effects of boreal forest clearcutting vary with riparian buffer width. Water Resour. Res. 61, e2024WR037705 (2025).
Wahli, T., Bernet, D., Steiner, P. A. & Schmidt-Posthaus, H. Geographic distribution of Tetracapsuloides bryosalmonae infected fish in Swiss rivers: an update. Aquat. Sci. 69, 3–10 (2007).
Mo, T. A. & Jørgensen, A. A survey of the distribution of the PKD-parasite Tetracapsuloides bryosalmonae (Cnidaria: Myxozoa: Malacosporea) in salmonids in Norwegian rivers – additional information gleaned from formerly collected fish. J. Fish Dis. 40, 621–627 (2016).
Gorgoglione, B., Bailey, C. & Ferguson, J. A. Proliferative kidney disease in Alaskan salmonids with evidence that pathogenic myxozoans may be emerging north. Int. J. Parasitol. 50, 797–807 (2020).
Philpott, D. et al. Effects of different preservatives during ecological monitoring of malacosporean parasite Tetracapsuloides bryosalmonae causing proliferative kidney disease (PKD) in salmonids. J. Fish Dis. 48, e14095 (2025).
Hutchins, P. R., et al. Exploration of the 2016 Yellowstone River fish kill and proliferative kidney disease in wild fish populations. Ecosphere 12, e03436 (2021).
Schager, E., Peter, A. & Burkhardt-Holm, P. Status of young-of-the-year brown trout (Salmo trutta fario) in Swiss streams: factors influencing YOY trout recruitment. Aquat. Sci. 69, 41–50 (2007).
Waldner, K. et al. A brown trout (Salmo trutta) population faces devastating consequences due to proliferative kidney disease and temperature increase: a case study from Austria. Ecol. Freshw. Fish 29, 465–476 (2019).
Szklo, M., Nieto, F. J. Epidemiology: Beyond the basics 3rd edn. 139 (Jones & Bartlett Learning, 2014).
Soomro, S. et al. Are global influences of cascade dams affecting river water temperature and fish ecology? Appl. Water Sci, 13, 106 (2023).
Carolli, M. et al. Impacts of existing and planned hydropower dams on river fragmentation in the Balkan Region. Sci. Total Environ. 871, 161940 (2023).
Fiske, P. et al. Novel large-scale mapping highlights poor state of sea trout populations. Aquat. Conserv. Mar. Freshw. Ecosyst. 34, e4067 (2024).
Lõhmus, M. & Björklund, M. Climate change: what will it do to fish-parasite interactions?. Biol. J. Linn. Soc. 116, 397–411 (2015).
Lauringson, M. et al. Spatial and intra-host distribution of myxozoan parasite Tetracapsuloides bryosalmonae among Baltic sea trout (Salmo trutta). J. Fish Dis. 46, 1073–1083 (2023).
Dash, M. & Vasemägi, A. Proliferative kidney disease (PKD) agent Tetracapsuloides bryosalmonae in brown trout populations in Estonia. Dis. Aquat. Org. 109, 139–148 (2014).
Sergeant, E. S. G. Epitools Epidemiological Calculators, Ausvet. (2018).
Debes, P., Gross, R. & Vasemägi, A. Quantitative genetic variation in, and environmental effects on, pathogen resistance and temperature-dependent disease severity in a wild trout. Am. Nat. 190, 000–000 (2017).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Hutchins, P. R., Sepulveda, A. J., Martin, R. M. & Hopper, L. R. A probe-based quantitative PCR assay for detecting Tetracapsuloides bryosalmonae in fish tissue and environmental DNA water samples. Conserv. Genet. Resour. 10, 317–319 (2018).
Hutchins, P. R., Sepulveda, A. J., Martin, R. M. & Hopper, L. R. Improved conventional PCR assay for detecting Tetracapsuloides bryosalmonae DNA in fish tissues. J. Aquat. Anim. Health 30, 164–170 (2018).
Klymus, K. E. et al. Reporting the limits of detection and quantification for environmental DNA assays. Environ. DNA 2, 271–282 (2020).
Merkes, C. et al. Reporting the Limits of Detection (LOD) and Quantification (LOQ) for Environmental DNA Assays: Data: U.S. Geological Survey Data Release (U.S. Geological Survey, 2019).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
Kuznetsova, A., Brockhoff, P. B. & Christensen, R. H. B. lmerTest package: tests in linear mixed effects models. J. Stat. Softw. 82, 1–26 (2017).
Fox, J., Weisberg, S., Price, B. car: Companion to Applied Regression. R package version 3.1-3 (2024).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer-Verlag, New York, 2016).
Wilke, C. cowplot: Streamlined Plot Theme and Plot Annotations for ‘ggplot2’. R package version 1.1.3 (2024).
Hartig, F. DHARMA: Residual Diagnostics for Hierarchical (Multi-Level / Mixed) Regression Models. R package (2024). version 0.4.7.
Lenth, R. emmeans: Estimated Marginal Means, aka Least-Squares Means. R package (2025). version 1.10.7.
link