Association between coffee and caffeine consumption and chronic kidney disease
Data collection and study population
The data utilized in this study are obtained from the National Health and Nutrition Examination Survey (NHANES). NHANES was conducted by the Centers for Disease Control and Prevention of America, which is a two-year-cycle cross-sectional survey and aims at evaluating the health and nutritional status of American population25. Initially, participants were interviewed at their home to gather essential background information, such as socio-demographic details, medical history, and family history. Later on, they attended a mobile examination center (MEC) to provide additional data such as anthropometric measurements, blood pressure, laboratory tests, and other relevant information.
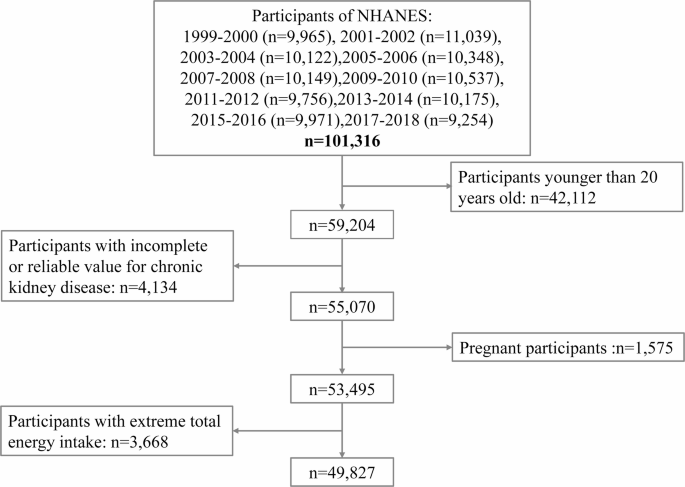
Ten cycles (1999–2000, 2001–2002, 2003–2004, 2005–2006, 2007–2008, 2009–2010, 2011–2012, 2013–2014, 2015–2016, 2017–2018) data included this study were downloaded from the NHANES website. A total of 101,316 participants from 1999 to 2018 enrolled in the survey. We excluded participants based on the following criteria: aged under 20 years old (n = 42,112), incomplete or unreliable CKD data (n = 4,134), pregnant (n = 1,575), and extreme total energy intakes (< 500 or > 8000 kcal/day for men, < 500 or > 5000 kcal/day for women) (n = 3,668)26. After exclusions,49,827 individuals were included in the final analysis (Fig. 1). NHANES was approved by the National Center for Health Statistics Research Ethics Review Board.

Flowchart of the screening process for the selection of eligible participants.
Chronic kidney disease assessment
CKD was defined as eGFR < 60 ml/min/1.73m2 or persistent albuminuria27. The eGFR was calculated using the 2021 chronic kidney disease-Epidemiology Collaboration Eq. (2021 CKD-EPI): eGFR = 142 × min(SCr/κ,1)^α × max(SCr/κ,1)^−1.209 × 0.9938^Age × 1.012 [if female], where SCr is serum creatinine in mg/dL, κ is 0.7 for females and 0.9 for males, α is −0.241 for females and − 0.303 for males, min denotes the minimum of SCr/κ or 1, max indicates the maximum of SCr/κ or 1, and Age is in years27,28. Albuminuria was defined as albumin-to-creatinine ratio ≥ 30 mg/g27,28. We did not define persistent proteinuria for most participants only measured once in the survey, and for the timing of second urine collection varied (first morning void vs. random) when albuminuria was repeated during selected cycles of NHANES29.
Exposure assessment
Frequency of coffee consumption, tea consumption, total caffeine consumption, caffeine from coffee, and caffeine from tea was obtained from two 24-hour dietary recall interviews, which were conducted by trained interviewers using an automated data collection system during the MEC examination30. Consumption of total caffeine and caffeine from tea and coffee was segmented into tertiles. Consumption of coffee and caffeine from coffee was divided into three categories. Participants with no consumption of coffee and caffeine from coffee were classified into group 1 (intake = 0), and individuals with consumption were divided into group 2 (< median) and group 3 (≥ median) based on its median intake (g/d) among the participants with consumption. Intake of tea and caffeine from tea was categorized into two groups. No consumption of tea and caffeine from tea was classified into group 1 (intake = 0), individuals with consumption were divided into group 2.
The measurement of urinary caffeine and its metabolite levels has been suggested as a reliable method for evaluating caffeine consumption31. Thus, we also evaluated the association of urinary caffeine metabolites and CKD using three cycles (2009–2010, 2011–2012, 2013–2014) data. All NHANES participants were required to provide urine samples in a mobile examination center (MEC). Ultra-high performance liquid chromatography-electrospray ionization-tandem quadrupole mass spectrometry was employed for the analysis of caffeine and its metabolites32including 1-methyluric acid (1U), 3-methyluric acid (3U), 7-methyluric acid (7U), 1,3-dimethyluric acid (13U), 1,7-dimethyluric acid (17U), 3,7-dimethyluric acid (37U), 1,3,7-trimethyluric acid (137U), 1-methylxanthine (1X), 3-methylxanthine (3X), 7-methylxanthine(7X), theophylline(13X), paraxanthine(17X), theobromine(37X), caffeine(137X) and5-acetylamino-6-amino-3-methy luracil (AAMU). Spearman correlation analysis revealed that all 15 metabolites exhibited a positive correlation with caffeine intake. Notably, 9 of these metabolites demonstrated a moderate positive correlation with caffeine intake (Spearman r, 0.501–0.578, P < 0.001) (Table S1). Therefore, we only assessed the relationship between 9 urinary caffeine metabolites and CKD. Bonferroni correction was applied to account for multiple comparisons, with a significance level of P < 0.05/9 for the nine metabolites assessed.
Covariates
Information of socio-demography, lifestyle, and health related factors had been collected by an interviewer using the Sample Person and Family Demographics questionnaires. Data included sex (male and female), age (20–39 years, 40–59 years, and ≥ 60 years), race (Mexican American, Other Hispanic, Non-Hispanic White, Non-Hispanic Black, and other races), educational level (below high school, High school, and above high school), marital status (married/living with partner, and widowed/divorced/separated/never married), body mass index (BMI) (normal: <25 kg/m2, overweight: 25 to < 30 kg/m2, obese: ≥30 kg/m2), physical activity (vigorous, moderate, and other), family income (Under $20,000, and $20,000 and over), smoking behavior (smoked at least 100 cigarettes in life or not), hypertension prevalence rate, diabetes mellitus prevalence rate, alcohol use (had at least 12 alcohol drinks/year or not), and total energy intake. We used the “dietaryindex” R package to calculate the AHEI based on dietary information33.
Statistical analysis
Statistical analysis was conducted using Stata 15.0 (Stata Corporation, College Station, TX). A new sample weight (the original 2-year sample weight divided by 2) had been constructed according to the analytical guidelines of NHANES when combing ten 2-year cycles of the continuous data34. In brief, we utilize the 4-year sample weights provided by NCHS instead of the 2-year sample weights. We conducted the Kolmogorov–Smirnov normality test to test the normality of continuous variables, and we described continuous variables with mean ± standard deviation (SD) (normally distributed) or median (interquartile range) (non-normally distributed). Student’s t-test (normally distributed) and the Mann–Whitney U test was used to examine the differences of continuous variable between the CKD group and the non-CKD group. Chi-square tests were chosen to examine the difference of categorical variables between the different groups.
In this study, we conducted binary logistic regression analyses to evaluate the association of coffee consumption, tea consumption, caffeine consumption and urinary caffeine and caffeine metabolite with CKD. Two models had been adopted to evaluate the association of coffee consumption, tea consumption, and caffeine consumption with CKD. Model 1, adjusted for age and sex; model 2 (fully adjusted model), adjusted for age, sex, race, marital status, educational level, family income, body mass index, physical activity, smoking status, alcohol consumption, hypertension, diabetes, alternative healthy eating index and energy intake. In addition, we performed subgroup analyses stratified by sex (male and female) and age (< 60 and 60~) to test sex and age as an interaction with coffee consumption, tea consumption, and caffeine consumption in the model that adjusted for the same covariates. As hypertension and diabetes may lie on the causal pathway between coffee/tea consumption and CKD, we conducted a sensitivity analysis excluding them. In addition, liner regression was conducted to evaluate the association of coffee consumption, tea consumption, and caffeine consumption with eGFR. A two-sided p < 0.05 was considered statistically significant.
link