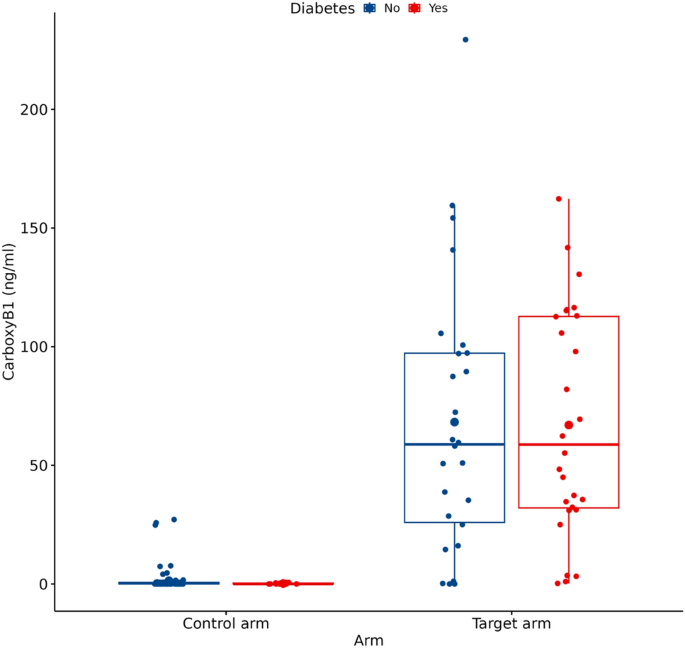
This study was performed at the University Hospital in Martin, Slovakia on a total of 142 subjects divided into a control group (n = 111) of nondialyzed subjects scheduled for elective invasive coronary artery angiography (ICA) and a target group consisting of 31 CKD G5d patients enrolled in the chronic dialysis programme at the Dialysis Center of the University Hospital in Martin. The first objective of the study was to measure the plasma concentration of CPB1 in CKD G5d patients and compare it to the mean CPB1 concentration found in controls. The second objective was to determine whether CPB1 concentrations are affected by selected parameters of dialysis adequacy and dialysis regimen. For each study participant in the target group, CPB1 was measured twice – at Time 1 and at Time 2—in that order, with a one-month—long gap between the two measurements. At Time 2, there were two new participants in the target arm. We included these two cases only in the descriptive statistical analysis. Regression analyses were conducted on paired samples in the target arm—therefore, the total number of cases for regression analyses performed in the target arm was n = 29, which is 58 observations in total. Patients in the control group were free of CKD G3b–G5 and/or acute kidney injury. However, there were several patients with CKD grades 1–3a. Subjects in the control group were not healthy controls since they carried significant burden of so-called civilization disorders. In each patient in the control arm, hemodynamically significant stenoses of coronary arteries were ruled out via the ICA procedure.
Informed consent statement
Authors declare that informed consent was obtained from all study participants and that all research was performed in accordance with the Declaration of Helsinki.
Explanation of study design
The design of this study takes into account the high morbidity of CKD G5d patients, in which nosologic entities belonging to the complex of civilization diseases, mainly cardiovascular diseases, play a major role. A control group consisting of completely healthy individuals would very likely create an artificial statistical contrast driven by the polymorbidity of the study participants on the side of the target arm and the disproportionately lower civilization disease burden on the side of the control arm. An ideal design would be proportionally equalized in terms of cardiovascular morbidity between target and control groups. Due to the relatively small target arm, it was impossible to have a target cohort in which each patient would undergo the ICA procedure and in which, compared to the control group, there would not be significant differences between the prevalence of disorders belonging to the complex of civilization diseases.
Invasive assessment of coronary arteries
Coronary artery angiography was indicated and performed in stable coronary artery disease patients in accordance to the ESC/EACTS Guidelines on Myocardial Revascularisation and ESC Guidelines for the Diagnosis and Management of Chronic Coronary Syndromes. Coronary artery stenosis was visually estimated by an experienced interventional cardiologist and was double–checked by another pair of eyes. Proximal LAD and left main stem stenosis of more than 50% were considered hemodynamically significant. In other coronary vessels, 70% narrowing was considered to be significant.
Dialysis procedure
The standard dialysis procedure as a part of the chronic dialysis programme was performed in the target arm of our study. In our dialysis center we utilized automated Fresenius 5008/5008S dialysis monitors paired with Fx80 high-flux dialyzers. In three cases Fx100 dialyzers were utilized. The KtV value was calculated every other month by the Online Clearance Monitor, which is the integral component of the Fresenius 5008/5008S automated dialysis monitor.
Laboratory procedures
All of the laboratory procedures were carried out in a certified medical biochemical laboratory. Plasma CPB1 concentrations were measured by the following ELISA kit, which was certified for experimental use:
Human Carboxypeptidase B1 ELISA Kit abx055595 (Abbexa LTD., Great Britain).
This ELISA kit is highly selective for its molecular target without known cross-reactivity with other carboxypeptidases. From each blood sample taken from the test subject plasma was immediately separated (15 min centrifugation at 3500 rpm). The plasma samples were immediately frozen at − 30 °C. The deep-frozen plasma samples were stored until the measurements were performed ad hoc for the entire ELISA kit. Each ELISA test was performed immediately after the samples were thawed. Repeating the freeze—thaw cycles was strictly prohibited. In the control group standard laboratory tests were performed, including measurements of creatinine and urea. Glomerular filtration was estimated according to the abbreviated Modification of Diet in Renal Disease (MDRD) formula.
Statistical methods
Statistical analyses were performed using IBM SPSS Statistics v. 25.0 software and R Statistics v. 4.0.5. The significance of differences between the control and target arms in the case of scale variables was assessed by the means of an independent samples t-test. In the case of violation of the homogeneity of variances assumption, a Welch–Satterthwaite correction was performed. Tests of two proportions were performed to determine the statistical significance of differences between the levels of binary variables.
To analyse the effect of predictors on the significant difference in the concentration of CPB1 between the target arm and repeatedly measured controls, a generalized linear mixed modelling technique (GLMM) was used. There was a large proportion of patients with zero CPB1 concentration among controls—therefore, we utilized GLMM with the zero-inflated gamma family with the log-link function to model the association between CPB1 and the predictors. The fitted model was then subjected to diagnostics by the quantile residuals. Marginal and conditional R2 values were used to quantify the goodness of fit. Intraclass correlation (ICC) was used to measure the proportion of total variability in the response that can be attributed to between-group differences. The fitted model was used to predict CPB1 for controls and for target arm cases and to obtain the estimated marginal means and post- hoc pairwise comparisons with the Benjamini–Hochberg adjustments of p-values. The estimated marginal means were visualized by an interaction plot with 95% confidence intervals (CI). From the standpoint of statistical power in a dataset with a relatively small number of observations, overcompartmentalization of the data caused by a large number of binary variables is undesirable. Therefore, we utilized a data averaging technique over a selected binary variable (Gender) to enhance the signal—to—noise ratio of the statistical information in the GLMM.
Linear mixed model regression (LMMR) was used to assess for associations between CPB1 and patient—specific variables, along with the dialysis regimen and dialysis adequacy parameters. The CPB1 variable was subjected to Hawkins and Weisberg power transformation. The variables included in the equation were Diabetes, Gender, Age, KtV value, amount of ultrafiltration performed per one week prior the dialysis session (UFw), water surplus prior the dialysis session (WS), number of dialysis sessions per one week-long dialysis cycle (Regimen), weekly duration of dialysis in minutes (MW) and dry weight (DW), which was measured via bioimpedance technique. The regression model was constructed using the Wilkinson–Rogers notation. The model was subjected to standard diagnostics using quantile residuals. Marginal and conditional R2 values were used to quantify the goodness of fit. Intraclass correlation was used to measure the proportion of total variability in the outcome variable that can be attributed to between-group differences. The model was used to obtain the back-transformed estimated marginal means and post- hoc pairwise comparisons with the Benjamini–Hochberg adjustments of p-values.
link